Sizing up cells: How stem cells know when to divide
A seemingly simple question has plagued scientists for more than a century: What controls the size of a cell? Just as body size may determine whether a football player is better suited to be a running back or a lineman, cell size shapes much of the function and behavior of the basic units of life. Cells of the same type maintain remarkably consistent sizes. In humans, red blood cells are uniformly small, while egg cells are among the largest cells in the body.
At Stanford University, postdoctoral researcher Shicong “Mimi” Xie studies how cells maintain precise control over their size. Xie and her colleagues examined whether cells in complex organisms regulate their size before dividing. In a recent Nature Communications paper, they showed that stem cells intrinsically control their size early in the cell division process.
To grow and divide, cells progress through the cell cycle, which includes four stages encompassing growth, DNA replication and division. In baker’s yeast, scientists have shown that cells must reach a critical size before progressing through the cell cycle to divide.
“If there’s some variation in cell size at the beginning of the cell cycle phase, that cell will delay entering the next cell cycle phase until it hits a threshold size,” Xie said.
Although some evidence suggests that cell size is regulated throughout the cell cycle in multicellular systems, other studies have found that this is not always true for mammalian cells grown in a dish. To address this controversy, Xie and her team set out to test whether the same principles apply in complex mammalian systems.
“I wanted to figure out if what was happening in budding yeast is still true of very complicated mammalian tissues,” she said.
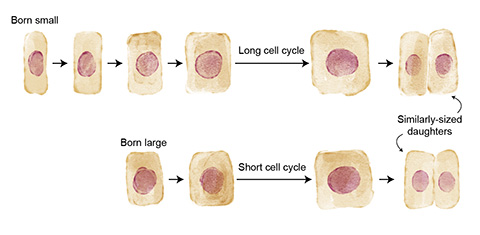
In a previous study, Xie found that mouse epidermal stem cells control their size before transitioning to the second phase of the cell cycle, similar to what occurs in baker’s yeast. Although she said she was surprised by the finding, Xie wanted to test whether the same phenomenon held up in other systems.
The scientists turned to a different organism: zebrafish. When zebrafish scales are removed, bone-forming cells called osteoblasts grow and divide to regenerate the tissue.
By analyzing live images of zebrafish scale regeneration, Xie found that osteoblasts also exit the first phase of the cell cycle and enter the second at approximately the same size. She observed the same pattern using light-sheet microscopy to image stem cells in mouse intestinal organoids, miniature cell clusters grown in the lab to mimic organs in the body.
Together, these results address the controversy surrounding cell size control in mammalian systems, showing that across living multicellular organisms, stem cells grow to a defined size before progressing through the cell cycle. The next question was how cells sense and enforce that size threshold.

To probe the mechanism behind this size checkpoint, Xie returned to her study of mouse epidermal stem cells and focused on the retinoblastoma, or RB, pathway, a central regulator of cell cycle progression that has long been suspected to link growth to division. She created mice that lack the RB protein RB1 and its paralog, RBL1, two closely related cell cycle regulators previously implicated in size control in human cells.
Without these proteins, mouse epidermal stem cells no longer waited to reach a target size before dividing. Instead, they progressed through the cell cycle based on time rather than size, effectively bypassing the size-control checkpoint.
These cross-species observations and the RB pathway experiments strengthen the conclusion that stem cells actively monitor their size before dividing, resolving longstanding questions about whether this form of size control operates in complex multicellular systems.
“Maybe the cells are not thinking about when they should become two cells, but the cells are really thinking ‘Have I grown enough to become two cells?’” Xie said.
While the study connects the RB pathway to cell size control, it does not yet explain how cells measure their own size or how RB activity is tuned to that measurement, leaving open important questions about the underlying molecular mechanisms.
Understanding those mechanisms may have implications beyond basic cell biology. As cells age, they often increase in size and function less effectively. Disruptions in size homeostasis are also a hallmark of diseases such as cancer. Determining how cells maintain size control could ultimately inform strategies to address age-related disease and tissue dysfunction.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

