The molecular orchestra of memory
What makes you remember a favorite vacation — the smell of the ocean, the taste of new foods — but forget where you left your keys? Inside the brain, the answer comes down to a carefully timed chemical choreography.
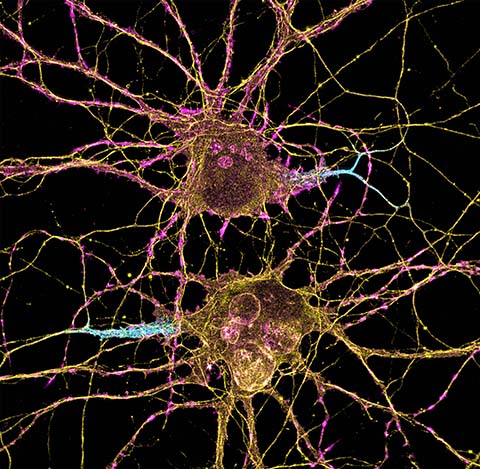
At its core, memory is biochemical. The brain’s ability to receive, store and recall information relies on ions, proteins and signaling pathways acting in concert to convert electrical activity into chemical changes at synapses and within neurons.
When this choreography falters, the consequences can be profound. Memory disruption underlies disorders such as Alzheimer’s disease, age-related cognitive decline and learning disabilities.
Among the key players in this molecular ensemble are calcium ions, known as Ca²⁺, the calcium-binding protein calmodulin, or CaM, and calcium and calmodulin-dependent kinase II, or CaMKII. Together, they form a signaling axis that drives the cellular changes required to form and maintain memories.

Neuroscientist A.J. Robison, professor in the department of physiology at Michigan State University, describes this signaling axis as fundamental to memory formation.
“We've known for more than 30 years that calmodulin functions as a calcium sensor and drives the activity of downstream signaling cascades, including CaMKII,” Robison said. “This activity is essential for the cellular correlates of learning, like long-term potentiation and long-term depression, and for actual learning itself in mice and other models.”
Yet this process is complex. Knowing the players is not the same as understanding the performance. Many questions remain about how these chemical changes produce lasting synaptic effects that support memories over days, years or even a lifetime.
Understanding these mechanisms advances basic neuroscience and points to potential therapeutic targets for disorders that impair learning, cognition and memory. Recent research published in American Society for Biochemistry and Molecular Biology journals is helping to clarify this signaling network, revealing new layers of regulation — and unexpected opportunities for intervention.
Calcium: A chemical spark
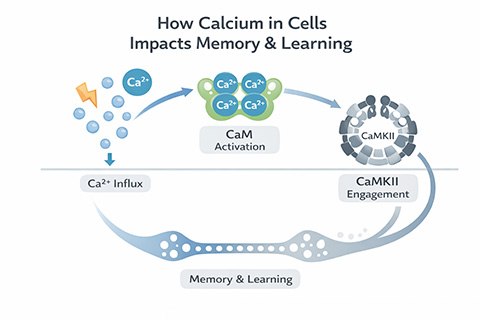
Every memory begins with a surge. Sensory inputs trigger rapid electrical spikes, called action potentials, that race along neurons and cause neurotransmitter release. Activation of the neurotransmitter receptors causes calcium ions to flood into the cell, acting as a spark for downstream signaling.
Repeated calcium surges signal that something worth remembering may be happening. Recurrent rises in intracellular calcium trigger long-term potentiation, a sustained strengthening of synaptic connections widely regarded as a hallmark of memory formation. Over time, these connections embed patterns of activity that encode memories.
Chiho Sugimoto, a postdoctoral fellow at Vanderbilt University who earned her Ph.D. in Robison’s lab, said this initial calcium spark is key to triggering the molecular cascade that underpins memory and learning. While calcium does not store memory itself, it sets the conditions that allow memories to form.
“Calcium is essential to how neurons communicate with each other by regulating the release of neurotransmitters and the strength of synapses, which are critical factors for learning and memory,” she said. “Calcium can control the timing, location and magnitude of synaptic signals through pathways such as CaM–CaMKII, which can shape neuronal circuits, synaptic plasticity and gene expression, the key drivers of brain function.”

A study published in the Journal of Biological Chemistry shows just how finely tuned that control can be. Using patch-clamp electrophysiology, a technique that measures electrical currents through specific ion channels, researchers studied voltage-gated ion channel activity in neurons. They focused on how retinoic acid, a vitamin A metabolite known to influence neural plasticity, modulates ion channel activity and synaptic function.
Researchers found that retinoic acid modulates neuronal firing by inhibiting potassium channels involved in repolarization and limiting calcium channel activation. By coordinating multiple ion channel targets, retinoic acid fine-tunes calcium entry into neurons, shaping neurotransmitter release and plasticity-related gene expression.
Vitamin A deficiency has been linked to cognitive impairments, highlighting how memory and nutrition may be connected. This work may help clarify how dietary or pharmacological interventions could support healthy brain function.
Calmodulin: The translator of calcium signals
A calcium pulse is only meaningful if the cell can understand it. This translation falls to CaM, which functions as calcium’s interpreter.
When calcium binds, CaM undergoes structural rearrangements that enable it to activate downstream enzymes, including CaMKII. In this way, CaM helps determine how neurons respond to activity patterns associated with learning, distinguishing background noise from signals strong enough to promote synaptic change.
CaM is not simply waiting for calcium to bind. CaM itself can also be chemically modified, and these posttranslational modifications can alter its activity and influence synaptic plasticity. Sugimoto said this layer of regulation may be critical for learning.
“CaM–CaMKII signaling is impacted by a variety of posttranslational modifications,” she said. “The discovery (of new CaM modifications would) add critical detail to our model of calcium effects on neuronal activity and synaptic strength.”
To this end, two complementary studies published in JBC highlighted the importance of a newly identified modification: acetylation.
One study identified acetylation as a novel CaM modification and found that it is more prevalent during active learning. Mice engineered to carry a form of CaM that could not be acetylated showed reduced learning and weaker fear-related memory. A second study identified SRC3 as the enzyme responsible for CaM acetylation. When SRC3 was blocked in mice, the animals had difficulty strengthening synaptic connections and showed weaker fear-based learning.
Together, the studies suggest that CaM acetylation plays an important role in synaptic plasticity and learning, acting as a regulatory layer that links neural activity to memory formation. The findings suggest that targeting CaM modifications such as acetylation could offer new strategies for preserving or restoring learning and memory in disease.
Sugimoto and Robison co-authored a commentary emphasizing the significance of these findings.
“What's novel about this is the discovery that calmodulin posttranslational modifications can drive differences in the activation of downstream pathways,” Robison said. “These studies help model how modification of calmodulin at these residues can drive differences in learning.”
Robison also emphasized the potential clinical implications of the findings.
“The SRC3 pathway could represent a (promising) approach to targeting CaM function in hippocampal neurons and may represent a novel pathway for therapeutic intervention in diseases of cognition, like Alzheimer’s or cognitive disabilities,” he said.
CaMKII: The molecular engine of memory
If calcium provides the spark and CaM interprets the signal, CaMKII does the heavy lifting. Once activated by calcium-bound CaM, CaMKII phosphorylates key synaptic proteins, reinforcing the connections that encode memory.
CaMKII has long been recognized as central to learning. However, precisely because of this central role, the enzyme has been viewed as an untouchable due to concerns that targeting it therapeutically could disrupt or erase established memories.
Recent studies published in JBC are beginning to address this uncertainty.
In one study, researchers temporarily inhibited CaMKII using the neuroprotective peptide tatCN19. Surprisingly, this short-term inhibition did not disrupt previously formed memories in mice, revealing unexpected stability in established memories.
This resilience is especially important when considering therapeutic applications. In pigs, temporary CaMKII inhibition reduced neuronal damage under physiological stress, pointing to a neuroprotective effect that could translate to human conditions such as recovery from stroke or cardiac arrest.
To understand how this level of regulatory precision is achieved, a second study from the same lab examined phosphorylation of CaMKII at a specific threonine residue. The researchers found that this modification is essential for multiple forms of long-term synaptic plasticity, highlighting how tightly regulated CaMKII activity is.
Together, these findings highlight the importance of precise CaMKII regulation for learning while suggesting that the CaMKII pathway can be safely modulated without compromising stored information.
Sugimoto sees this regulatory balance as central to human health.
“CaM-CaMKII signaling is a central molecular mechanism for learning and memory that allows us to translate our experiences and environment into synaptic changes that encode long-term memories,” she said. “Dysfunction in this signaling is implicated in diseases like mood disorders, schizophrenia, epilepsy and autism spectrum disorder, indicating that understanding how this signaling is regulated can provide us with novel therapeutic strategies.”
The future of fine-tuning memory
The same chemistry that allows you to remember the details of a vacation also determines whether everyday moments are lost to distraction. Calcium sparks, CaM binding and CaMKII reinforcement together help determine what stays and what fades.
Because of the critical nature of this axis and the ubiquity of some of its components, developing therapeutics remains challenging. Among these players, CaM has been especially difficult to target because of its widespread presence in the body.
However, recent research suggests that targeting specific regulatory nodes along the axis may be a promising alternative.
“CaM is ubiquitous and plays a role in so many cell types and functions that targeting it therapeutically would doubtless have myriad off-target effects,” Robison said. “If we can target specific CaM modifications or target this CaMKII pathway in specific neurons in the brain through SRC3, maybe therapeutics won't have the detrimental effects that you might have by targeting calmodulin directly.”
As researchers continue to untangle how calcium signals are interpreted, translated and reinforced in neurons, the picture of memory formation is becoming clearer. These insights bring the field closer to strategies that could improve and preserve learning and memory while protecting the brain, ensuring that the delicate chemical choreography underlying memory remains in step.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

