How copper delivery fuels bacterial respiration
Copper is an essential metal that functions as a cofactor for many enzymes, including cytochrome C oxidase, or Cox, which drives cellular respiration. Mechanisms of copper uptake, trafficking and efflux from cells must be tightly regulated to maintain energy production, as excess copper can generate toxicity and cellular damage. Aerobic bacteria are well-studied models of cellular respiration. However, the specific pathways by which copper is properly delivered to Cox enzymes remain unclear. Likewise, while most bacteria rely on an adenosine triphosphatase, or ATPase, known as CopA, to export copper, not all ATPases participate in detoxification, and coordination with chaperones and periplasmic proteins for copper delivery to enzymes is not fully understood.
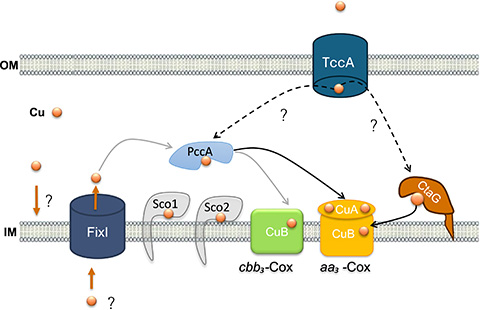
Hala Kasmo of the University of Namur and an international team published an article in the Journal of Biological Chemistry identifying the roles of several proteins in copper homeostasis in Caulobacter vibrioides, or C. vibrioides, an aerobic bacterium that has two Cox enzymes, cbb3-Cox and aa3-Cox. They demonstrated that while C. vibrioides lacks a CopA-like ATPase for detoxification, it encodes a FixI ATPase involved in copper transport.
To determine whether FixI participates in detoxification or copper delivery to Cox enzymes, researchers created bacterial strains lacking FixI, exposed cells to excess copper and measured both cell growth and cbb3-Cox and aa3-Cox activity. They found that deleting FixI did not affect cell growth or survival under high copper conditions, demonstrating that FixI is not required for copper detoxification. The activity of cbb3-Cox, however, was strongly reduced in the FixI-deleted strain, indicating that FixI is involved in copper delivery to cbb3-Cox.
To investigate how copper is delivered to aa3-Cox, researchers performed a genetic screen on a transposon-mutagenized library in a strain lacking both FixI and cbb3-Cox. This screen revealed that TccA, an outer membrane receptor, is required for aa3-Cox activity. They also demonstrated that while cbb3-Cox activity requires the periplasmic chaperone PccA, aa3-Cox activity depends on both PccA and CtaG, an inner membrane protein.
These findings highlight that C. vibrioides uses diverse pathways to supply copper to its two terminal oxidases, independent of detoxification. To further understand these mechanisms, researchers can screen molecules that selectively inhibit FixI, TccA or PccA and measure the effects on cellular respiration, which can inform future antimicrobial strategies, such as disrupting copper delivery in pathogens.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

