When things get SAPpy: Novel insights into complement
The body uses two distinct arms of the immune system to effectively fight off various pathogens, such as bacteria and viruses. The innate immune system is considered the “first responder” and includes the complement system. This system is a key regulator of inflammatory signal amplification and clearance of dead or damaged cells to manage pathogenic infections.
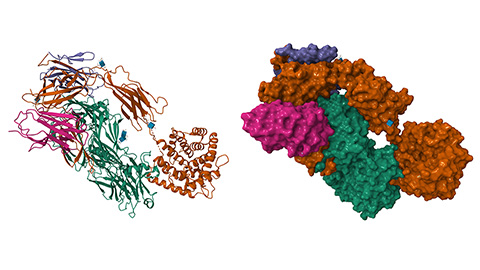
C4BP is a binding protein responsible for the inactivation of complement protein C4b and, consequently, the complement cascade. Interestingly, C4BP is also known to form higher-order structures, or HOS, allowing C4BP to interact with other complement-related proteins. Recently, Tereza Kadavá and a team of scientists at Utrecht University characterized the interactions of C4BP HOS with two of its known binding partners, C4b and serum amyloid protein, or SAP, and identified potential areas of crosstalk between the two binding interactions. The study was published in Molecular & Cellular Proteomics.
Using a combination of mass photometry and high-speed atomic force microscopy, the team determined that C4b formed three different types of complexes with C4BP HOS particles. The most abundant binding conformation consisted of a 1:1 C4BP:C4b binding ratio, while smaller quantities of complexes were bound in ratios of 1:2 or 1:3. Additionally, they determined that these interactions are specifically mediated by C4BP-α chains within the C4BP structure.
Conversely, SAP showed a strong preference for 1:1 binding ratios, which led to the discovery of SAP pentamer engagement with the entire C4BP HOS. By using cross-linking mass spectrometry, Kadavá and her colleagues discovered that SAP interacts with the C4BP HOS via the C4BP oligomerization core. The researchers also noticed that even in the presence of abundant C4BP-α, C4BP-SAP complex formation did not occur, suggesting that this process is mediated separately from C4BP:C4b binding.
This work provides novel proteomic insights into the complement cascade and structurally defines the C4BP-C4b complex for the first time.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

