Catching tau in the act
Tau acts as a stabilizer for the brain’s highways, reinforcing neuronal microtubules that ferry nutrients and other essential cargo.
But, tau can go rogue, detaching from these highways and forming toxic aggregates with itself and other proteins, disrupting their function. These tangles are linked to cognitive decline and neurodegenerative diseases, including Alzheimer’s disease.
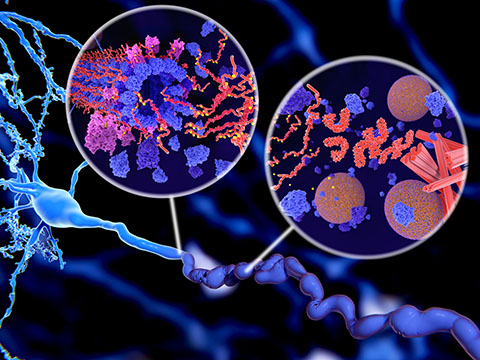
The molecular partnerships tau forms after detachment, and how they drive disease, have been difficult to identify.
In a recent Molecular & Cellular Proteomics paper, Emory University researchers developed a method to capture these tau interactions. Using a proximity-labeling technique that tags proteins only when tau molecules meet, the team uncovered hundreds of tau-associated proteins also found in diseased human brains.
The work tackles a long-standing challenge in tau research. Traditional methods for identifying protein interactions rely on extracting intact complexes from cells, but tau aggregates are often insoluble material that these methods miss.
Sarah Shapley, a former graduate student from the lab of Nick Seyfried, professor in the department of biochemistry and neurology at Emory, is the first author on the paper.
“The lab wanted to understand interactors of insoluble proteins, but traditional methods get rid of these interactions during sample prep,” Shapley said.
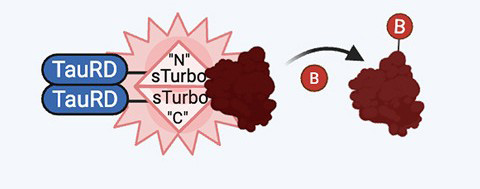
To get around that problem, Shapley and colleagues turned to split TurboID, a biotin-labeling enzyme that becomes active only when its two halves come into close proximity. By attaching each half to the region of the tau protein known to be involved in the interactions that lead to aggregation, the researchers created a system that labels neighboring proteins specifically when tau molecules interact and oligomerize.
Shapley likens the approach to a forensic dye pack.
“It’s like someone robbing a bank and the ink explodes,” she said. “Anyone nearby gets marked. Later, you can go back and ask, ‘Who was in the room when this happened?’”
Using this system in human cell models, the team identified more than 700 tau-associated proteins. While some were expected, like microtubule regulators, others stood out — especially RNA-binding proteins involved in splicing, nuclear transport and mRNA stability.
“We assume that tau is a neuronal microtubule-binding protein, but maybe it has a strong, noncanonical role in RNA splicing, RNA binding and even regulation of translation,” Seyfried said. “These interaction studies reveal interactions that point to novel biology.”
The researchers compared the tau interactome in cells with large-scale proteomics datasets from postmortem human brain tissue. Many of the same RNA-binding proteins appeared in brains affected by Alzheimer’s and other neurodegenerative diseases, suggesting relevance to human pathology.
“Even in this very reductionist cell model, we were capturing interactions that translated to what we see in human brain tissue,” Shapley said.
The findings shift attention to earlier, transient stages of tau pathology — before large multi-protein tangles form — a timing that could be crucial for therapy development.
“A lot of neurodegeneration research has focused on the insoluble protein fibril rather than the soluble toxic oligomeric species,” Shapley said. “As we see more of this in the therapeutic space, I’d be interested to see how an oligomeric tau is targeted.”
Beyond tau, the authors note that the split TurboID strategy is a major advance, one Seyfried plans to use to study other aggregation-prone proteins involved in neurodegenerative diseases.
“This proximity labeling approach has expanded our ability to probe aggregate-prone proteins,” Seyfried said. “This can reveal new groups of interactors that could be modifiers of disease.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

