Itching for answers about the role of lipids in psoriasis
While we often dwell on the importance of the heart and brain, the human skin is the largest organ in the body and vital to our survival. It serves as a barrier to water, guarding the body against dehydration, temperature extremes and harmful chemicals. Lipids in the skin are critical to its function as a water barrier since fat and water don’t mix.
Christopher Thomas is a lecturer and researcher at the School of Pharmacy and Pharmaceutical Sciences at Cardiff University whose research focuses broadly on oxidized lipids and their role in human health and disease, particularly in the skin. Thomas became interested in the skin when he was pursuing a Ph.D. in chemistry and working on developing topical treatments for rheumatoid arthritis.
“We were trying to put fatty acids and fish oils through the skin,” Thomas said.
Bigger research questions arose for Thomas regarding skin physiology and how products are metabolized once they enter the skin. His postdoctoral studies then focused on lipoxygenase, or LOX, enzymes within platelets in blood. Thomas’ current research is a marriage of his previous work; he now investigates the role of LOX enzymes in the skin.
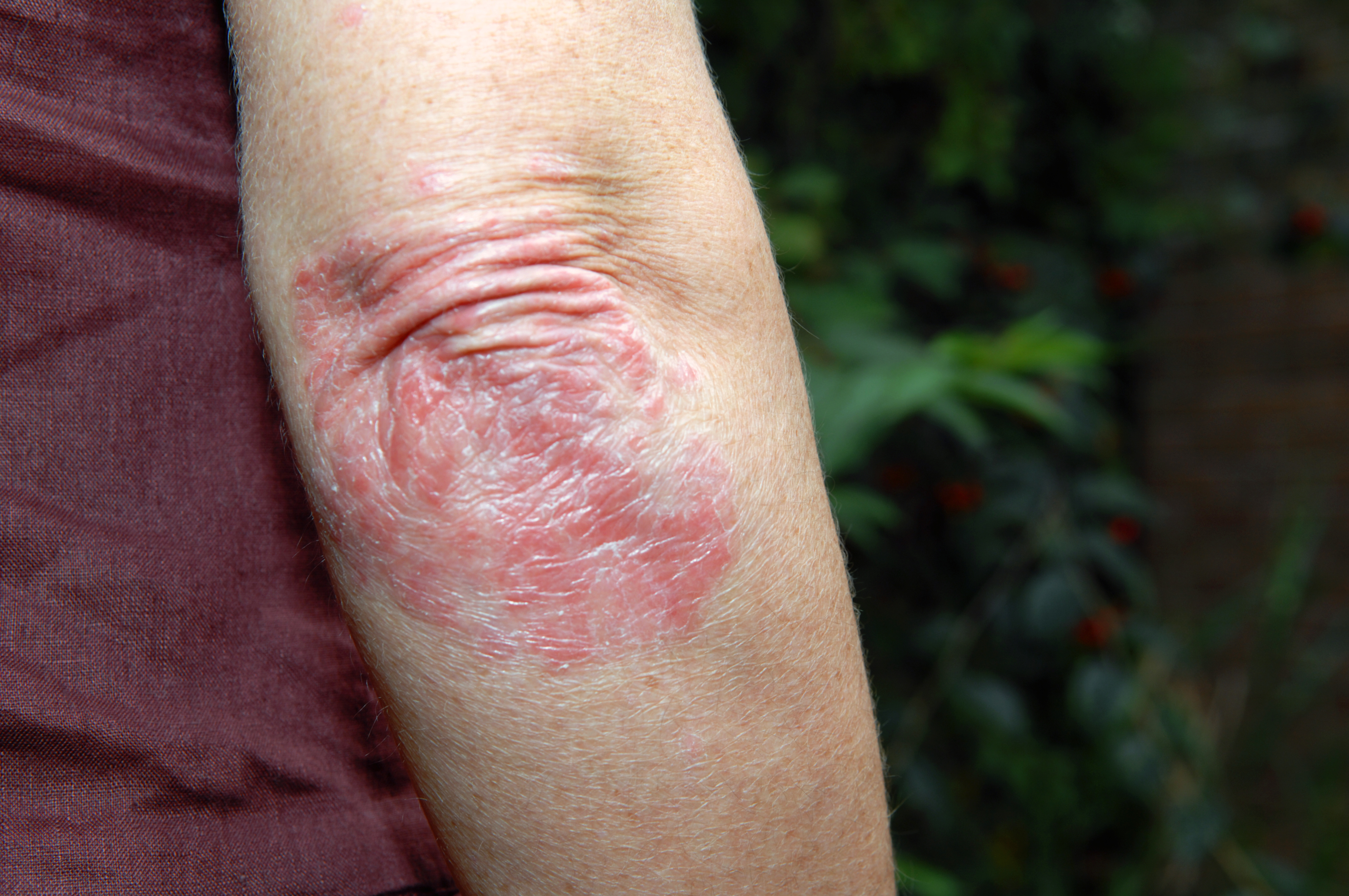
In a recent paper in the Journal of Lipid Research, Thomas and collaborators describe studying a biosynthetic pathway in the skin that is vital to formation of the complex mammalian skin barrier. In this pathway, ceramide lipids containing a fatty acid, linoleic acid, are oxidized by LOX enzymes. Ceramides of varying carbon lengths are oxygenated by LOX. Following some intermediate steps, a final hydrolysis step yields a terminal product called 9,10,13-trihydroxy-10E-octadecenoic acid, which has been found at higher than normal levels in patients with eczema.
Researchers know little about the exact location in skin layers of these intermediate and terminal species and their roles in health and disease. Thomas developed methods to study the quantity and location of these species, using liquid chromatography with tandem mass spectrometry. Method development for any kind of lipid system can be quite challenging, he said, because the lipids are repelled by any solutions containing water.
As part of this research, the team analyzed human psoriatic skin samples and showed that the linoleic acid oxygenation pathway was highly dysregulated. Unoxidized intermediates in the pathway were elevated significantly relative to nonpsoriatic skin samples, and oxygenated species were reduced.
The researchers also used a publicly available data set from the Gene Expression Omnibus to compare the gene profile of skin from psoriatic lesions to that of unaffected skin in patients with psoriasis. They found that the genes for the entire biosynthetic pathway for oxygenated species described here are expressed more in the skin from human psoriatic lesions. This suggests that the regulatory machinery to control even unaffected skin lipid metabolism is abnormal in psoriasis patients.
Thomas’ next avenues of research will use an exciting new addition to the lab: a 3D printer to generate novel skin models. This is a project shared with several other researchers at Cardiff who are interested in studying printed skin from a variety of research angles.
“It’s a collaborative feel where we’re all interested but we’ve all got our slightly different hats on,” he said.
Thomas added that he also views 3D-printed skin as a way to advance the goal of producing representative human healthy and diseased skin models, which could help to reduce animal testing in the pharmaceutical and cosmetics industries.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Glaucoma model links immune signaling to disease progression
Researchers at Duke University determine genetic variations that could increase the risk of developing glaucoma.

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

