How mucus keeps us healthy
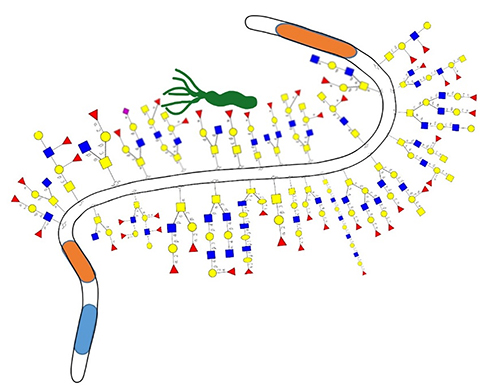
We tend to associate mucus with colds and flu, but its role in the body is complex and varied. This colloidal network of salts, enzymes, antibodies, glycans and glycoproteins is found in the nose, mouth, lungs, stomach, intestines and reproductive tract. The slimy mucus layer of the stomach includes bottle brush–shaped glycoproteins called mucins that contain carbohydrate chains called glycans that help build a selective, viscous barrier between epithelial cells and the external environment.
Sara Lindén at the University of Gothenburg in Sweden studies interactions between microbes and mucosal surfaces in the stomach, intestine and airways. “Until relatively recently many researchers thought the role of the glycans on the mucins was merely to hold water and thereby create a lubricating barrier on our mucosal surfaces,” Lindén said. “Recently, a number of studies have shown that the mucin glycans have roles in binding to and governing interactions with microbes, including both the natural commensal microflora and disease-causing microbes.”
Helicobacter pylori is a common bacteria that can cause harmful infection. Spread through contaminated drinking water, poor sanitation and saliva sharing, it might be present in as much as 50% of the world’s population. Most people are unaffected, but some experience stomach pain and develop ulcers. Chronic infection can lead to certain stomach cancers, and many strains of H. pylori are now antibiotic resistant.
Lindén’s doctoral studies involved H. pylori interactions in the human stomach, and she has continued this line of research. A recent article in the journal Molecular & Cellular Proteomics reports on novel glycan structures her team found in mucin from stomach samples of individuals with and without H. pyloriinfection.
The lab’s previous studies showed people benefit from mucins that bind efficiently to Helicobacter, as this limits the bacteria that come into contact with human cells and cause disease. Proteins on the H. pylori surface called adhesins bind to surface structures on the cell, Lindén explained. “Mucins can also bind to these adhesins and act as releasable decoys that Helicobacter can bind to instead of the human cell.”
Lindén and her team were surprised at the diversity they found in over 600 carbohydrate structures identified from just 28 patient samples. Patients infected with H. pylori showed larger interpatient variability in this diversity than noninfected patients. In this context, Lindén said, “The high diversity of glycans suggests that interactions with microbes in the stomach can vary enormously between different individuals.”
Previous experiments in animals have shown that H. pyloriinfection and mucin production are cyclically related: Infection leads to decreased mucin production as well as changes in the mucin glycosylation, allowing more H. pylori to bind cells and infect. In this study, Lindén’s team found specific structures that correlated particularly well to H. pylori binding.
All this could inform the design of glycan-based therapeutics as an alternative to the standard use of multiple antibiotics. Gurdeep Chahal, lead author of this article, imagines that “targeting the H. pylori adhesins with the analogues of these glycans could reduce the bacterial colonization in the stomach while simultaneously treating chronic gastritis.”
Lindén sees the results going further: “The work presented here shows how structures in the human body differ between individuals and how this affects interactions with a pathogen … part of a larger concept of understanding how our body surfaces defend themselves against disease causing microorganisms and how we can enhance these defense systems to treat and prevent infections without antibiotics.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

The molecular orchestra of memory
Calcium, calmodulin and calcium/calmodulin-dependent kinase II form a molecular axis that turns fleeting neural activity into lasting memories. New research shows how memories are stabilized, and possibly even protected or repaired.

Differences in pili structure modulate bacterial behavior
Researchers demonstrate how small changes in the structure of hair-like protein appendages can affect the behavior of Acinetobacter bacteria.

Cholesterol regulatory genes predict liver transplant outcomes
Researchers identify a link between cholesterol-regulating genes and liver transplant success, which could improve donor screening and patient outcomes.

