How a tiny pet store fish became the center of neuroscience research
I spent the better half of my twenties peering at tiny little fish under the microscope, and it was one of the most exciting time of my life.
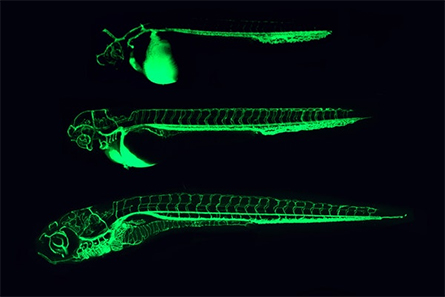
Every morning, I would rush to the lab to see if my fish had laid eggs. I watched the brain cells of these completely transparent organisms multiply under the lens. I still remember the first time I saw a live neuron grow in front of my eyes, in the brain of a young fish larva. This striped tropical fish could fit in the palm of my hands, and yet is one of the most important organisms in biology, allowing researchers to answer fundamental questions in neuroscience, developmental, cancer, disease and regenerative biology.
The beginnings of zebrafish research can be traced back to early 1930s when Charles Creaser at Wayne State University in Detroit began using zebrafish (Danio rerio) eggs to show students the development of a live embryo and the movement of blood inside its arteries. Creaser was able to do this because zebrafish females release eggs from their body which are fertilized by the sperm released from the male. The freshly formed embryo is accessible to the observer from the moment it is fertilized. The fact that zebrafish embryos and larvae are transparent means studying the internal parts of the animal is a breeze. Creaser established methods for rearing, feeding and breeding zebrafish in the lab, but widespread use of the animal did not take off for another three decades.
Like Mendelian genetics, zebrafish had to be rediscovered. In the late 1930s, George Streisinger and his family moved across the Atlantic in an attempt to escape the anti-Semitic living conditions of 20th century Hungary. Streisinger’s passion for science got him a job under Myron Gordon, one of the fish researchers at the New York Zoological Society.

Streisinger dedicated almost two decades of his career to studying the genetic code of viruses that infect bacteria, called bacteriophages. He used these viruses to decipher the way genetic material is coded in living beings. Despite having made such fundamental contributions to science using phages, Streisinger wished to switch to a different system. He aspired to understand the developmental of the nervous system in vertebrates, especially how neurons find their partners and make connections. At this time, in early 20th century, animals such as medaka fish and goldfish were the go-to choices for researchers interested in understanding development. But Streisinger went another way. “The story goes that he walked into a pet store in Oregon, and asked the owner, ‘what’s a good simple vertebrate that I could study?’ and the owner pointed at zebrafish. At least that’s what fishlore says,” recounts Karuna Sampath, a professor studying control of embryo development at Warwick Medical School.

Having already worked with zebrafish at the Zoological Society in New York, he was aware of the immense potential the fish held for biology experiments. When he joined the University of Oregon in 1960, Streisinger jumped headfirst into generating mutant fish strains. His initial lab was a converted army barrack, inside which he and his colleague Charlene Walker reared zebrafish in tanks. Zebrafish are naturally found in tropical climates of South Asia and can only survive in temperatures from 24°C-38°C, which meant that Streisinger and Walker had to get creative in maintaining the temperature of the hut at a sweet spot. In summer, water was poured on the roof to keep things cool, while in winter, electric heaters kept the hut toasty. Things soon started to pick up. In the two decades that Streisinger dedicated to zebrafish research before his death, he not only worked on deciphering neuronal development in the fish brain, but also set up techniques for linking mutations in the fish genome to changes in the fish’s appearance.
Streisinger’s goal of bringing zebrafish up to par with model systems of the likes of Drosophila and rodents was carried to fruition by the army of colleagues he had inspired. Zebrafish’s ease of maintenance and breeding, year round supply of embryos, external fertilization giving access to embyros from hour zero, and transparent young stages of development makes observation easy. But to really put the animal over the top, it was important to show that it was amenable to mutations, a key tool for biologists.
That’s exactly what Christiane Nusslein-Volhard at the Max Planck Institute in Tubingen and Wolfgang Driver at Massachusetts General Hospital did. They spearheaded the “Big Screen,” spread across multiple labs over the two continents, to produce about 4000 fish strains, each of which had a different aspect of early development disrupted. The result of this massive project was published as a collection of 37 papers in a single issue of the journal Development, finally announcing to the academic world that zebrafish was here to stay. On the heels of this accomplishment came the sequencing data of the entire zebrafish genome in 2001, which gave the community the ability to pick out the genes and match them to the changes observed in the big screen fish mutants.
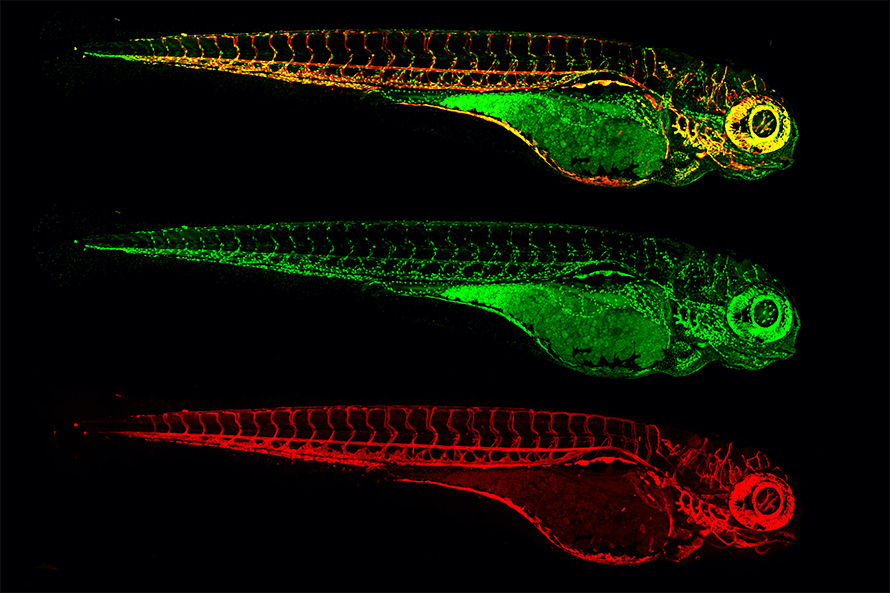
What began as an animal picked up from the local aquarium is now an established vertebrate model system used across thousands of labs to answer questions about the formation of the nervous system, evolution of behaviors, development and treatment of cancers, regeneration of tissues and much more. One can actually look at the activity patterns of every cell in the entire zebrafish brain while the fish swims around. You can screen thousands of candidate drugs by just dumping them into the water with the fish and watching what happens.
“It’s a good balance of being simple enough to get a holistic understanding of mechanisms underlying behaviour, while at the same time sharing a lot of conserved similarities in terms of anatomy, genes, molecules, that you can link to mammals and to humans”, says Caroline Wee, a neurobiologist lab at the Institute of Molecular and Cell Biology, A*STAR Singapore.
Julien Vermot, who runs a lab at the Department of Bioengineering, Imperial College London uses these fish for something unique. To study the effects of mechanical forces on the development of the heart, students in his lab stop the zebrafish heart from beating for as long as a couple of days. It’s one of very few animals that can survive this. Despite such unique advantages, the model isn’t everyone’s first choice and certainly not as popular as mice or rats. As Vermot puts it, “It’s a fish. And we are not fish. So, you need to convince people that what you do is meaningful for the entire community.” Wee adds, “You can argue they’re similar, but it’s hard to mentally draw the link between fish and humans.”
While the connection between zebrafish research and humans may not be obvious, the benefits for them are. Rita Fior, group leader at the Champalimaud Centre for the Unknown has shown how these animals can be used to test chemotherapy treatments for cancers growing in actual human patients. Fish injected with cancer cells from patients and given the same treatment as the patient show 84% similarity in the response to the drugs. This sets up precedence for potentially screening drug combinations in fish to look at their efficiency in reducing growth of cancerous cells before administering them to patients.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

