From the journals: JLR
A molecule to recycle LDL. Reviewing sortilin’s role in disease. New mice help track PLD expression. Read about papers on these topics published recently in the Journal of Lipid Research.
A molecule to recycle LDL
Low-density lipoprotein, or LDL, sometimes is known as bad cholesterol. An elevated level of circulating LDL increases risk of myocardial infraction and stroke. Thus, the body must be able to clear LDL.
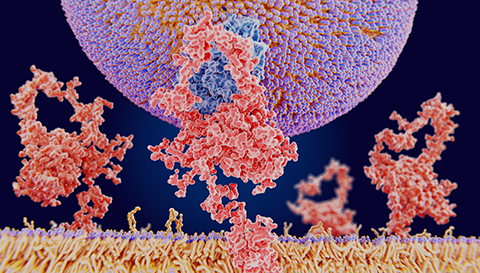
The LDL receptor, or LDLR, molecule plays a major role in LDL clearance by binding to apolipoprotein B, a component of LDL, thus triggering intake, or endocytosis, of circulating LDL. The LDL then goes to a lysosome for hydrolysis, and the free LDLR is inserted back into the plasma membrane. This process is called recycling.
A number of proteins regulate LDLR expression and recycling. The small GTPase RAB10 is one of these, and RAB10 also is required for LDL uptake. Deleting RAB10 decreases endocytosis of LDL but increases accumulation of transferrin proteins, which play a role in iron metabolism in mammals.
Small GTPases cycle between inactive and active states. This molecular switching recruits effector proteins to membranes. Researchers have reported that RAB10 has a role in membrane trafficking in various cell types. And scientists know that RAB10 mediates insulin-stimulated transport of vesicles containing the glucose transporter GLUT4 to the plasma membrane.
Taslima Khan and a team at the University of Michigan recently investigated the role of RAB10 in LDLR and transferrin receptor, or TFR, recycling. Their study was published in the Journal of Lipid Research.
In this study, the group cited evidence that RAB10 promotes recycling of both LDLR and TFR: RAB10 depletion results in a decreased ratio of both receptors on the membrane without changing its expression and increased accumulation of both receptors in the cytoplasm, and RAB10 is colocalized with both receptors. The researchers suggest that RAB10 plays a broad role in trafficking, however, and thus targeting RAB10-mediated LDLR recycling is of limited therapeutic value.
Reviewing sortilin’s role in disease
Receptor-associated protein, or RAP, is a molecular chaperone that plays a role in folding and processing low-density lipoprotein, or LDL, receptors. Researchers have associated the glycoprotein sortilin, one of the binding partners of RAP, with Alzheimer’s, prion and Parkinson’s diseases and high cholesterol. Also, according to human genomewide association studies, the gene that encodes sortilin is associated with LDL cholesterol. However, researchers do not yet understand the mechanics of this association.
That association and the sortilin–RAP interaction suggest that sortilin may have a role in lipoprotein trafficking. Some studies have reported that sortilin regulates very low-density lipoprotein cholesterol secretion from hepatocytes, thus regulating LDL cholesterol levels. By extension, these findings suggest that sortilin affects cardiovascular and metabolic diseases.
Kelly Mitok, Mark Keller and Alan Attie at the University of Wisconsin–Madison have reviewed the literature, focusing on sortilin’s sometimes confusing role in cardiovascular and metabolic diseases while outlining its structure, expression and regulation. Their review was published recently in the Journal of Lipid Research.
New mice help track PLD expression
Phosphatidic acid, or PtdOH, is a phospholipid that plays a role in membrane fusion of secretory vesicle and granules. This negatively charged signaling molecule is recruited into microdomains, promotes membrane curvature and induces conformational changes in proteins. PtdOH interacts with proteins to regulate exocytosis in neuroendocrine cells and is involved in synaptic vesicle recycling in the mammalian central nervous system. It can be synthesized by phospholipase D, or PLD, mediated hydrolysis of certain phospholipids.
Researchers who study PLD activity often see conflicting results in the localization and expression of isoforms PLD1 and PLD2 because the reagents used in these experiments are unreliable. To address this problem, Casey Barber and a team at the Johns Hopkins University School of Medicine genetically altered mice to express labeled PLD1 and PLD2. Using brain slices and neuron cultures from these mice, the researchers investigated expression and localization of the isoforms.
Their results, published in the Journal of Lipid Research, showed that while PLD1 expression is predominant in neurons, PLD2 is expressed mostly in glial cells. The researchers also observed that overexpression of PLD1 reduces complexity of dendritic arborization and that PLD1 inhibition increases dendritic branching. Moreover, they found that PLD1 is expressed in the retina, although they do not know its role there. Overall, the group learned about the role of PLD in neurons and in glial and retina cells. And their genetically altered mice can be used for future studies of localization and expression of different tissues.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Glaucoma model links immune signaling to disease progression
Researchers at Duke University determine genetic variations that could increase the risk of developing glaucoma.

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

