Dysregulation of a lipid transfer protein linked to brain disorders
Advanced studies of human genetics are a big wave in the medical sciences. Collaborative teams of clinical geneticists and bioinformaticians are surfing this wave, rapidly discovering genomic variations associated with specific human disorders. This trend is providing scientific bases for personalized medicines but also new, important questions linked to the basic biochemistry field.
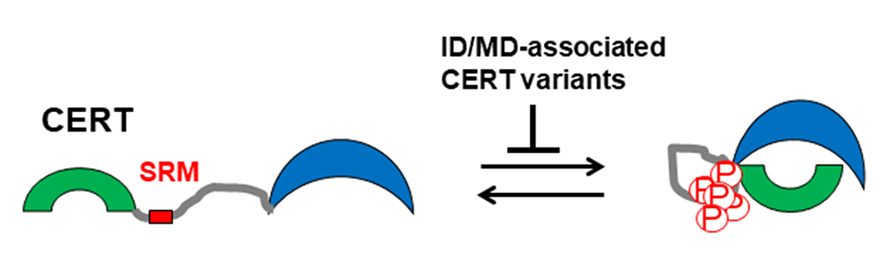
Ceramide transport protein, or CERT, moves the waxy lipids known as ceramides in cells for the synthesis of sphingomyelin, a membrane lipid that is ubiquitous in mammalian cells. In 2007, researchers found that CERT is functionally repressed by multiple phosphorylations of a serine-repeat motif, or SRM, in CERT. At the time, scientists regarded this finding as pure biochemistry of a protein.
However, a decade later, large-scale human genetic studies on intellectual disabilities and mental development disorders, or ID/MD, showed that missense mutations in or near the CERT SRM-encoding regions are associated with a type of autosomal dominant hereditary ID/MD. The dominant inheritance was in line with a prediction from the previous biochemical study that loss of hyperphosphorylation of the SRM renders CERT abnormally active.
Our recent collaborative study confirmed this prediction by demonstrating that substitution of a serine residue in the SRM with other residues similar to variants found in ID/MD patients results in dysregulation of CERT in cultured cells. Nonetheless, several ID/MD-associated missense mutations that occurred in the CERT gene CERT1 also are mapped outside the SRM. This riddle was answered by another recent study showing that a non-SRM variant also compromises the SRM hyperphosphorylation, thereby abnormally activating CERT.
Moreover, cell biological analysis showed that abnormally activated CERT mutants exhibit an aberrant punctate distribution in cells, suggesting that the subcellular distribution pattern is applicable as a diagnostic tool to assess whether a CERT1 variant is an abnormally activated type that may cause ID/MD, although the precise identity of the puncta structure remains undetermined.
Want more lipid research news?
Check out Lipid Trends, a curated collection of hot picks from the world of lipid research, brought to you by LIPID MAPS.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

