From the journals: Brain Awareness Week
It’s Brain Awareness Week. Here’s a special edition of “From the Journals” featuring recent brain research published in the American Society for Biochemistry and Molecular Biology’s open-access journals: Journal of Biological Chemistry, Journal of Lipid Research and Molecular & Cellular Proteomics.
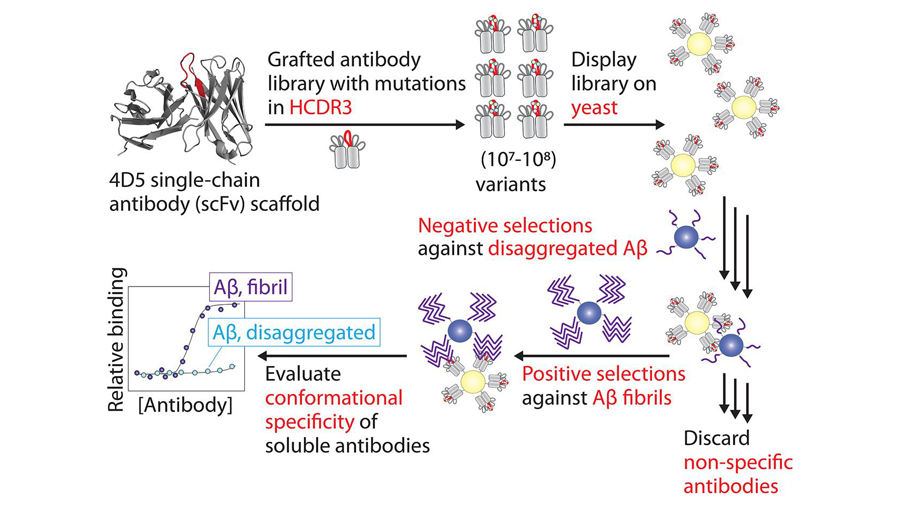
Anti-amyloid antibody design — a possible treatment for Alzheimer’s
Antibody design for the treatment of disease historically has been a challenging field to break ground on due to their (large) size, heterogeneity, hydrophobicity, low solubility and low kinetic stability. In 2017, companies Biogen and Eisai developed two clinical trials for an antibody treatment called Aducanub; given intravenously to patients, the treatment’s goal was to attack the beta-amyloid plaques that are common in Alzheimer’s. These monovalent antibodies — that is, antibodies with an affinity for one antigen — work together to probe for plaques with multivalent antigens to bind to, signaling for their destruction. Recently, a research group designed antibodies to be bivalent, hoping to increase binding to amyloid plaques. In their systematic approach to antibody library design and selection of conformational antibodies, they isolated anti-amyloid antibodies with high affinity and specificity; they also identified robust methods for future generation of a range of different types of amyloidogenic aggregates. The work was published in the Journal of Biological Chemistry.
Potential biomarker in Alzheimer’s disease progression
N-linked glycosylation is the process in which a glycan (carbohydrate) undergoes many reactions to attach to a nitrogen atom in eukaryotic cells. This often is associated with cell migration and protein stability. It has been known that mutations in the genes responsible for this process result in changes to the glycan, many of which are linked to a variety of diseases. Recent advances in proteomic and glycoproteomic liquid chromatography–mass spectrometry techniques have led to analysis of glycans found in cerebral spinal fluid. One particular study looked at the cerebral spinal fluid of Alzheimer’s disease patients, noting global changes to detected N-linked glycans. Read more about how this might be a breakthrough potential biomarker in Alzheimer’s progression in the journal Molecular & Cellular Proteomics.
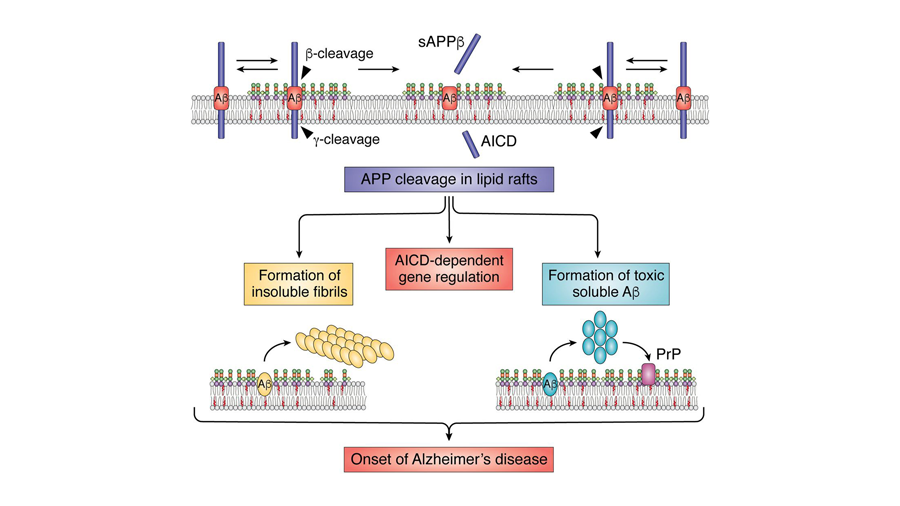
Lipid rafts and neurodegeneration
The cell membrane is highly dynamic in its composition. This means that not all areas of the cell membrane will contain the exact same type and amount of specific lipids. Thus, lipid rafts are defined best as small areas within the cell membrane that are rich in similar glycolipids, sphingolipids and cholesterol; these areas are dynamic and transient, meaning they also can change throughout a cell’s life cycle. Researchers believe that these lipid rafts organize in discrete manners to determine the function of cellular activity. In the brain, they have been linked to signal transduction and homeostasis, therefore playing roles in the brain’s performance. A 2020 thematic review in the Journal of Lipid Research explores how alterations of lipid rafts have been associated with a decline in brain performance, which in turn could play a central role in many neurodegenerative diseases.
Parkinson’s and vesicle trafficking
Synaptic vesicle trafficking is a unique type of cellular trafficking — which generally requires that vesicles carrying the appropriate cargo bud from a source membrane, travel in the appropriate direction and then fuse with the proper cellular target. Studies of alpha-synuclein have correlated it to the synaptic vesicle pool maintenance, yet the precise functions of α-synuclein are still unknown. Being an intrinsically disordered protein, α-synuclein has exhibited multiple discrete membrane-binding modes. In this 2019 review in the Journal of Biological Chemistry, scientists revisit what is known about both α-synuclein’s and other intrinsically disordered proteins’ functions in normal neuronal cells, such as synaptic vesicle trafficking, and discuss how these functions are known to change in disease states such as Parkinson’s disease.
Parkinson’s at the chromosome level
The leucine-rich repeat kinase 2, also known as LRRK2, is a well-studied gene that encodes a complex protein with both kinase (which transfers phosphate from ATP to other molecules) and GTPase (which converts GTP to GDP) domains. Interestingly, many studies have linked mutated variants of the LRRK2 gene in patients to a predisposition for the development of late-onset Parkinson’s disease. The most common mutation in the LRRK2 gene results in a change from glycine to serine at amino acid 2019, which is the highest known risk factor for developing both genetic and sporadic Parkinson’s to date. In a recent study in the Journal of Biological Chemistry, researchers found that LRRK2 controls the directional motility and chemotaxis of macrophages (immune cells that eat damaged cells and activate inflammation) in Parkinson’s, suggesting a molecular pathway for therapeutic targeting.
Parkinson’s disease — a possible treatment?
As of yet, there is no cure for or therapy to reverse the damage caused by Parkinson’s disease. Most preclinical trial therapeutics target restoring dopamine synthesis, neuroprotection, genetic neuromodulation and/or addressing disease-specific pathogenic variants, with many of these targets being drug-based. A new, alternative approach that researchers are exploring is the transplantation of human PSC–derived purified CNTN2+ progenitors (a very specific type of stem cell). With this transplantation, researchers reported enhanced dopamine release from transplanted cells in the host brain and alleviation of phenotypes related to Parkinson’s disease in animal models. To acquire these stem cells, researchers must grow and purify the cells and thus need molecular targets to verify a successful purification; these markers also were identified, proving purification of the stem cells to be suitable. Read more about their work in the journal Molecular & Cellular Proteomics.
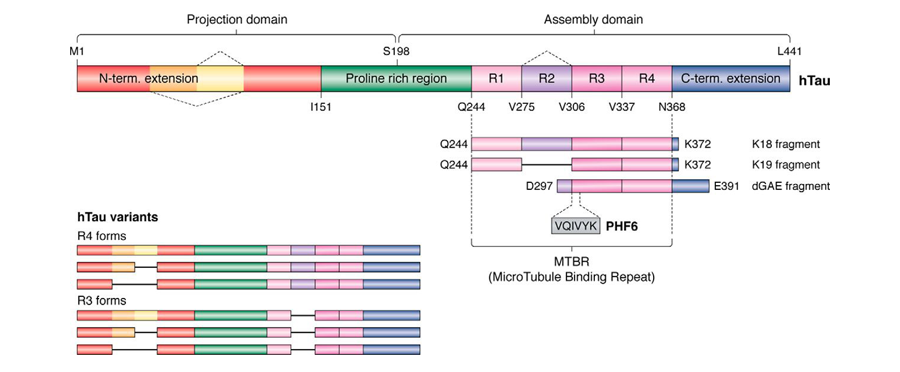
Better identifying dementia variants by tau proteins
While some forms of dementia present with some molecular signatures, such as Lewy body figures, hydrocephalus or vasculitis (to name a few), this is not true for all subclassifications. Before the advancement of cryo-electron microscopy, research on correlations of the tau protein and forms of dementia were seemingly inconclusive, leading some scientists to relate tau buildup broadly to neurodegeneration. In a recent review article in the Journal of Biological Chemistry that addresses advancements in cyro-EM for use in brain research, researchers urge scientists to revisit tau structure databases, specifically for tau variants, as they were able to distinguish tau variants within various forms of dementia.
Prions and neurodegenerative disease
Prions are a type of pathogenic material that can cause a specific type of brain proteins, prion proteins, to misfold. When this occurs, the misfolded proteins often cause various prion-related diseases that result in inflammation, damage and death to brain cells. Interestingly, the normal proteins contain a receptor for amyloid-beta, and therapeutic targets to this receptor’s domain potentially could prevent prion infection. Efforts to make these aptamers have been hindered due to nucleic acid stability, specificity and delivery across the blood–brain barrier. In a 2022 Journal of Biological Chemistry review article, researchers address each of these concerns and the potential for aptamers as therapeutics for both prionlike diseases and many other neurodegenerative disease with similar domain targets.
Creutzfeldt–Jakob disease
Creutzfeldt–Jakob disease is prion based and causes rapid degradation of brain cells, which is ultimately fatal. It can be caused by genetics, sporadically or by exposure to the prionlike infectious agents. The hallmark of the disease is the mutated isoform of the prion protein — PrPc — that has been infected with prions, yielding a scrapie, PrPsc. The detailed mechanism of this infection and subsequent mutation has yet to be elucidated fully, but researchers have broken ground in this area with the discovery of the N-terminal polybasic region, or NPR, of the protein. Of note, their data suggested that both the amino acid sequence of the NPR and charge contribute to the conversion of PrPc to PrPsc. This work was published in the Journal of Biological Chemistry.
Prion proteins and mass spectrometry
Preclinical trials of many therapeutics targeting PrPsc are in the works. Most target the normal protein, PrPC, with hopes of decreasing its levels in the brain so individuals are less susceptible to prion infection and protein mutation. To quantify lower levels for proof of concept, molecular assays such as ELISAs have been used, but these types of assays could miss vital concentration differences in the cell. Thus, another research group recently developed a mass spectrometry multiple reaction monitoring system of nine PrP tryptic peptides; these peptides are quantified relative to an isotopically labeled recombinant protein standard for human samples, and they proved to be a suitable method for biomarker detection. The work was published in the journal Molecular & Cellular Proteomics.
Potential biomarkers for ischemic stroke
Despite the frequency of stroke as a cause of death worldwide, the identification of key molecular biomarkers for ischemic stroke is still elusive. Recent advancements in high-throughput omics, combined with other sources of data integration, have identified a network of 18 genes and proteins that are expressed differentially at a very early stage after stroke in mouse brains, two hours after an ischemic stroke event. Researchers found that both GADD45G and CTNND2 were common to mouse and human stroke victims and could be used in stroke diagnosis and treatment planning. For more information, read the study in the journal Molecular & Cellular Proteomics.
Diabetes, phospholipid profiles and stroke
Diabetic patients are about six times more likely to suffer from a disabling stroke than people without diabetes. This is due to the vulnerability of diabetes patients to developing carotid endarterectomy artery plaques as part of their disease, which leads to a higher risk of ischemic stroke. To understand better the phospholipidic profiles of diabetes patients, a research team recently analyzed the phospholipid composition of those plaques. The team found choline-ethanolamine phosphotransferase 1, a protein known for its biosynthesis of a subclass of phospholipids, to be upregulated, suggesting that arachidonoyl phospholipids are more likely to be abundant in diabetic patients and thus opening up possibilities for future targets and treatment. The work was published in the Journal of Lipid Research.
Multiple sclerosis biomarkers
Multiple sclerosis most often is associated with demyelination (damage to the myelin sheath covering of neurons) and progressive loss of axons (the fibers that carry impulses away from neurons). Normally, MS has one of two distinct clinical presentations — active or inactive — that physicians deduce based upon the patient’s symptoms. Researchers are interested in discovering molecular biomarkers of the different stages of MS in hopes of uncovering its molecular machinery and potential targets for therapeutic treatment. Understanding that MS causes neuroinflammation and neurodegeneration, which often are associated with sphingolipid species, a new study in the Journal of Lipid Research looked for sphingolipid biomarkers for MS disease and pathology.
Traumatic brain injury and Parkinson’s disease
There is no FDA-approved drug for treatment of traumatic brain injury. Many omics techniques have attempted to uncover the pathology of the disease, with hopes of also uncovering possible therapeutic targets. With technological advancements allowing for high-throughput research methods, the TBI landscape now can be explored at the microproteomic level. Specifically, one research group combined in silico docking experiments and in vitro experiments to conduct a spatiotemporal study of mild TBI injury. From this they deduced the plasticity factor of the brain to be molecular, lying within the proteomic level, thus supporting clinical data that TBI could be a predisposition for Parkinson’s. You can read more about their findings in the journal Molecular & Cellular Proteomics.
Mitochondria and traumatic brain injury
The primary injury of TBI often results in cognitive and motor deficits due to the pathology of mechanisms such as oxidative stress, inflammation and neurotransmitter imbalances. The secondary mechanisms of TBI often are observed to persist over days and weeks. One example of a secondary mechanism is mitochondrial dysfunction, in which damaged mitochondria no longer are able to play their usual role in autophagy. Recently, researchers found that, with a TBI injury, the activation of a protein called acid sphingomyelinase occurs post-transcriptionally and results in mitochondrial impairment that promotes neuroinflammation and further brain-function defects. Their study was published in the Journal of Lipid Research.
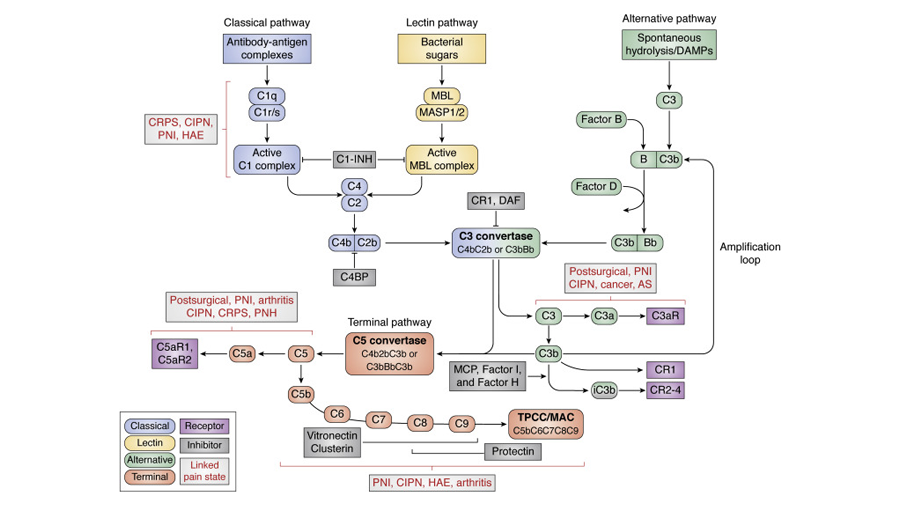
The complement cascade and neuroinflammation
The complement cascade, a mechanism of the innate immune system, is the first response for recognition and repair of damaged and infiltrated tissues of the body. While its general mechanism is thought to be well characterized, there are gaps in knowledge of its underpinnings in interactions with nervous system tissues and neuronal processes surrounding development, aging and disease. While its upregulation may be beneficial in some cases, such as clearance of infiltrated tissues by a pathogen or plaque buildup from aging, upregulation also demonstrates issues concerning chronic inflammation, pain and overall neural dysfunction. Thus, this review in the Journal of Biological Chemistry sets out to consolidate the understanding of communication between the immune system and neurons while posing future topics of discussion, such as time-dependent considerations for complement activation, homeostasis and modulation, and the limitations of study in animal models.
Dopamine transporter and addiction
Dopamine is a type of neurotransmitter that is used to modulate motion, cognition and the reward system in vertebrates. Dopamine signaling occurs through the dopamine transporter, or DAT, a component of the presynaptic network. For proper function, DAT often interacts with scaffolding proteins via its C-terminus PDZ domain-binding sequence. Disruption of the PDZ domain causes changes to the expression and distribution of DAT at the synapse that, in turn, alter DAT signals. When dopamine signaling is altered, it is associated with many neuropsychiatric disease pathologies, such as schizophrenia, attention-deficit hyperactivity disorder and addiction. In a recent mouse model study published in the Journal of Biological Chemistry, researchers compared presynaptic and postsynaptic adaptions to this DAT-scaffold protein interaction and not only confirmed the role of the PDZ domain on DAT proteins but also found that it resembled the changes seen in addicted brains, opening up further investigation of dopamine homeostasis and compulsive drug intake.
Linking chronic migraines and opioid-induced hyperalgesia
The prolonged use of opioids to treat migraines can result in a condition called opioid-induced hyperalgesia, which can lead to increased pain and headaches. Recently, proteomic researchers hypothesized the overlap to be due to neuropeptide dysregulation at the molecular level. In their label-free, nonbiased liquid chromatography–mass spectrometry analysis of more than 1,500 peptides, they identified the pituitary adenylate cyclase-activating polypeptide as a link between chronic migraines and opioid-induced hyperalgesia in the larger pain-processing pathways, laying the foundation for future studies of therapeutic approaches to treat migraines. Their work was published in the journal Molecular & Cellular Proteomics.
TRESK and migraines
Both TRESK and TREK-2 are genes coding for monomers of two-pore domain potassium channels. These subunits together provide for a potassium current to be generated in primary sensory neurons. When the TRESK subunit endures a frameshift mutation, neuronal hyperexcitability occurs, which has been linked to migraines. In a recent paper in the Journal of Biological Chemistry, this effect was shown to be enhanced by the addition of T2A3, a selective activator for the TREK-2 subunit. The researchers who conducted the study propose T2A3 as an emerging drug target of interest for treating migraines.
Neural development and lipids
Ethanolamine phosphotransferase 1, or EPT1, is the enzyme responsible for transferring phosphoethanolamine from a cytidine diphosphate-ethanolamine metabolite to a lipid acceptor in the process of producing ethanolamine glycerophospholipids (a specific family of lipids). Historically, no research has probed into mutations of EPT1 on production and concentration of ethanolamine glycerophospholipids as a result of disease. A new study in the Journal of Lipid Research, however, connects EPT1 to the production of ether-linked plasmalogen [1-alkenyl-2-acyl-glycerophosphoethanolamine (plasmenyl-PE)], essential in neurodevelopment of neuronal membranes and myelin sheaths as well as homeostasis in ether-linked phospholipids.
Presynaptic plasticity and synaptic repair
During development, growth-associated protein 43, also known as GAP43, is found in the axons of almost all neurons. This protein is involved in the formation of presynaptic terminals, synaptic plasticity, axonal growth and axonal regeneration. In addition, the developing brain contains urokinase-type plasminogen activator receptor, also known as uPAR, which binds urokinase-type plasminogen activator, or uPA, to convert plasminogen into plasmin — a protein that breaks up blood clots. During the conversion process, cell-signaling pathways that promote cell migration, proliferation and survival mechanisms are activated. Historically, this mechanism was not known to be preserved or well understood in maturing brains, and GAP-43 was located sparsely at presynaptic terminals and axonal growth cones. New research, however, reveals that some brain injuries cause the release of uPA in neurons, triggering axonal growth and synaptic formation. In addition, researchers recently revealed a novel role of uPA in activating the synaptic vesicle cycle by presynaptic recruitment and activation of GAP-43 in cerebral cortical neurons. Read more about uPA/uPAR binding affecting the underlying mechanism of presynaptic plasticity and synaptic repair in the Journal of Biological Chemistry.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

