
Bertrand Coste and the pressure receptor
The 2021 Nobel Prize in physiology or medicine was awarded to David Julius and Ardem Patapoutian for their discoveries of receptors that sense temperature and pressure, work that exemplifies how difficult research can be. Through hundreds of mice, tens of thousands of cells and millions of bacterial colonies, the research groups that made the winning discoveries persisted in asking important questions about how the brain detects its surroundings.
Researchers in Julius’ and Patapoutian’s labs who made key discoveries at the bench worked through many technical problems and disappointments in pursuit of the molecules behind sensation. “The Secret History of Touch” tells five stories of their persistence. Here is the fourth.
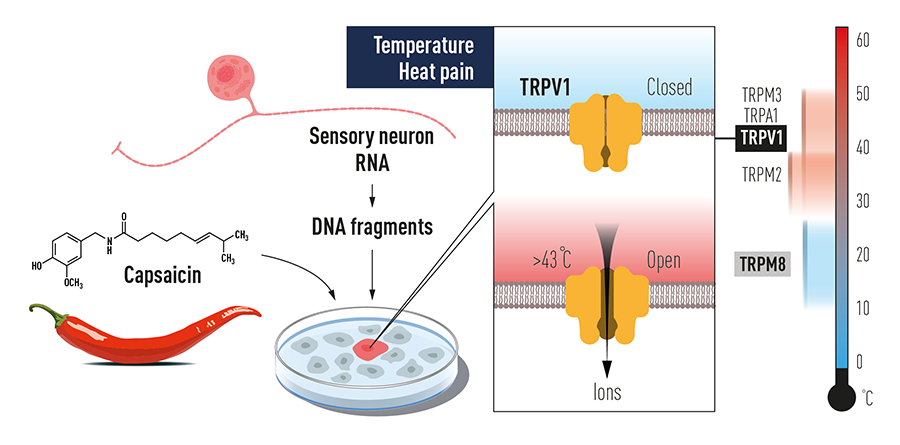
"He struck gold"
In the mid-2000s, Ardem Patapoutian, known for his studies of temperature sensitive ion channels called TRPs, wanted to transition into studying mechanotransduction.
Patapoutian did not have time for an interview for this story, but in an email, he wrote, “After studying temperature sensation for 10 years, it was a natural transition to ask how mechanical force is sensed.”
Like the search for the cold receptor, it was another risky question, fraught with the possibility of failure. Sanjeev Ranade, who joined Patapoutian’s lab as a graduate student to study thermosensation, said, “Many labs had been looking for the identity of the gene or genes that allow us to sense touch. Many labs were not successful in finding these genes.”
So when physiologist Bertrand Coste joined the lab as a postdoc to search for the protein that lets human neurons sense pressure, Ranade said, “It was one of those almost ‘I can’t believe you’re doing this’ kind of projects.”
Like Makoto Tominaga in David Julius’ lab, Coste was an experienced electrophysiologist who came to a new lab in California with a reputation for receptor hunting and immediately began to build a new electrophysiology rig. He had studied mechanosensory neurons as a graduate student and was skilled at recording from a neuron with an electrode while gently prodding it with a tiny glass probe — a technique that few others in the world used.
“Recording these mechanically activated currents naturally pushed me to the question of what are the ion channels that are involved in this activity,” Coste said. Patapoutian’s lab, which lately had cloned TRPA1 and TRPM8, struck him as the ideal environment to try to answer the question.
Coste initially planned to sift through the DRG to find neurons that respond to mechanostimulation and then extract their RNA and screen for the genes governing pressure sensation. But pilot experiments made it clear that the strategy was impractical. So he took a different approach: He tested as many cell lines as he could lay hands on, looking for one that would respond to pressure by depolarizing. “With immortalized cell lines you have endless materials,” he said. “It’s easy to use, and every cell is a clone of the other cells.”

Coste found a neuroblastoma line that fit the bill and winnowed down its list of highly expressed genes to the most interesting candidates: transmembrane proteins with unknown function that shared some characteristics with ion channels.
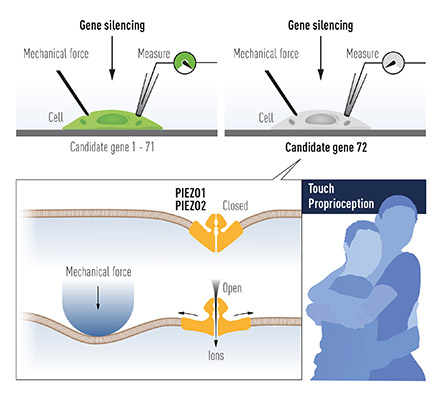
That left a list of dozens of genes. Coste began to use siRNA to knock them down one at a time and then prodded the altered cells, watching for one that would fail to respond to mechanical stimulation. Electrophysiology is highly reliable but also very slow. He averaged about two candidate genes a week, and, for a year or more, every single candidate he tested had no effect on the cells’ response. Not making progress can be tough. A few months in, after many trials and no success, Coste said, “My mood was declining very fast.”
“We are scientists. We like to think, to problem solve,” he said. While planning the screen and working around the technical issues, he’d had a chance to solve problems. “But the screen itself was very repetitive.” Coste brought the problem to Patapoutian, explaining that even though he was excited about the possible results of the project, the mindless grind of doing the same experiment over and over again and getting no result was difficult. Patapoutian, according to Coste, was understanding; he assigned a technician to help and suggested a second, more tractable project he could work at on the side. “I was relieved of doing every day the same thing, every day having negative results,” Coste said.
Finally, after ruling out 72 candidate genes, Coste found an siRNA that blunted the cell’s response to mechanical stimulation. Ranade said, “They could have easily quit at 72, and said, ‘OK, it’s been a year, we have nothing.’ And yet, they found it at 73.”
The gene was mysterious, but it was large, with an estimated 24 to 36 transmembrane domains. Besides controlling the neuroblastoma cells’ mechanical response, when cloned into mechanically inert cells, it made them sensitive. The team dubbed it Piezo, after the Greek word for pressure, and soon found that mice have two closely related Piezo proteins.
Coste said characterizing the channel was exhilarating: “After one year of screening … every experiment you do is telling you something interesting on the activity of this channel.”
Coste and many colleagues in the Patapoutian lab would go on to show that Piezo1 and Piezo2 play integral roles in mammals’ sense of touch and many other physiological functions. Even before that, Ranade said, it was clear that the finding was important. “Every single one of us in the lab, when Bertrand found Piezos, we all kind of knew … that he struck gold.”
Motivation to persist
What keeps researchers going through failure after failure? ASBMB Today asked psychologist Ayelet Fishbach, who studies motivation, work and learning from failure.
Fishbach and colleagues recently published a study investigating work-related one-time failures. After taking a two-option multiple choice quiz, Fishbach and colleagues found, participants who had received negative feedback performed worse on a follow-up quiz than those who had received positive feedback.
Although the study investigated one-time failure to guess an insignificant piece of information rather than the longer and more fraught process of research, Fishbach said that its conclusions reflect her own experience as a researcher and mentor.
“There are two categories of problems with learning from failure,” she said. Cognitively, it can be harder to learn from the unexpected than the expected and more difficult to seek out the reasons that a hypothesis missed the mark than to accept that it was correct. “Getting people to pay attention to what didn’t work is notoriously hard,” she said.
The second category is emotional. It is easier to ignore a failure than to engage with it. And sometimes people — especially those with relatively little expertise in a field — learn the wrong lesson from failure, concluding that they are unable to execute the task at hand. What’s more, Fishbach said, “Doing something with no sense of progress is psychologically hard. You can tell yourself that one day it will pay off, but it’s not today or yesterday or tomorrow. And people give up.”
Much like David Julius, Fischbach said that social support, encouraging words and help with troubleshooting from others can help keep people motivated. But, Julius pointed out, “It’s not just me. They have to have their own gyroscope.” He added that there are times when he has doubted the future of a project and has gotten inspiration from persistent trainees.
The Secret History of Touch Series
Michael Caterina and the capsaicin receptor
How the Julius lab found that an ion channel senses heat
Makoto Tominaga, Toby Rosen and TRPV1 heat sensation
Nobelist’s postdoc searches for a receptor for mint and cold
Patapoutian’s postdoc unearths the powerful Piezo gene
Bertrand Coste and the pressure receptor
Nobelist’s lab pins down pressure sensing in mice
Seung Hyun Woo, Sanjeev Ranade and Piezo2 in the sense of touch
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

