Stabilizing the enzyme in fish odor syndrome
Fish odor syndrome, or trimethylaminuria, is a disease in which the liver cannot break down the smelly chemical trimethylamine, or TMA, that is produced by enzymes from bacteria residing in the gut. There is no cure for fish odor syndrome, which gives people an unpleasant fishy smell that can affect breath, sweat, urine and vaginal fluids.
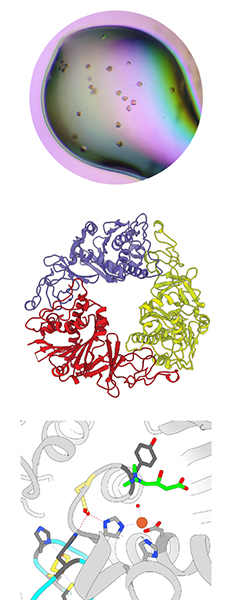
Our research team at the University of Warwick is working to prevent the syndrome through studying the enzyme in the gut that produces trimethylamine.
Fish odor syndrome starts when an enzyme pathway in the gut called CntA/B produces TMA. The enzyme breaks down a TMA precursor called L-carnitine, which is found in dairy, fish and meat. If an individual lacks a functional liver enzyme called FMO3, they cannot degrade TMA into a non-smelly chemical form, trimethylamine oxide, or TMAO. The TMA then builds up in the body and ends up in bodily fluids.
In a recent paper, published in the Journal of Biological Chemistry, our team in Yin Chen's lab at Warwick's School of Life Sciences focused on the CntA protein of the CntA/B enzyme, to stabilize and study it.
CntA/B is a notoriously hard enzyme to study, but once it was stabilized, we were able to gain insight into how CntA perceives its L-carnitine substrate with a 3D crystal structure model, and by studying the complete electron transfer pathway, we could see how the protein is able to turn over TMA.
Now that we understand how exactly TMA is produced in the gut and that the enzyme can be inhibited, there are grounds for further research into future discovery of drugs targeting the TMA-producing enzyme in the human gut.
We have identified novel, drug-like inhibitors that can inhibit CntA function and thus TMA formation with the potential to attenuate TMA formation in the gut microbiome. This is vital not only for people who have fish odor syndrome, but also because TMA can accelerate atherosclerosis and heart disease.
This article was adapted from a University of Warwick press release. Read the original here.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Glaucoma model links immune signaling to disease progression
Researchers at Duke University determine genetic variations that could increase the risk of developing glaucoma.

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

