Early lipid changes drive retinal degeneration in Zellweger spectrum disorder
Zellweger spectrum disorder, or ZSD, is a rare genetic disorder that disrupts essential cellular processes in infancy and worsens over time, often leading to blindness and life-threatening complications. Individuals with mild ZSD typically live 30–40 years, though treatment is limited to symptom management. This disorder disrupts essential cellular processes by impairing peroxisomes — small organelles responsible for breaking down toxic substances and producing lipids, which are vital for brain and organ development.
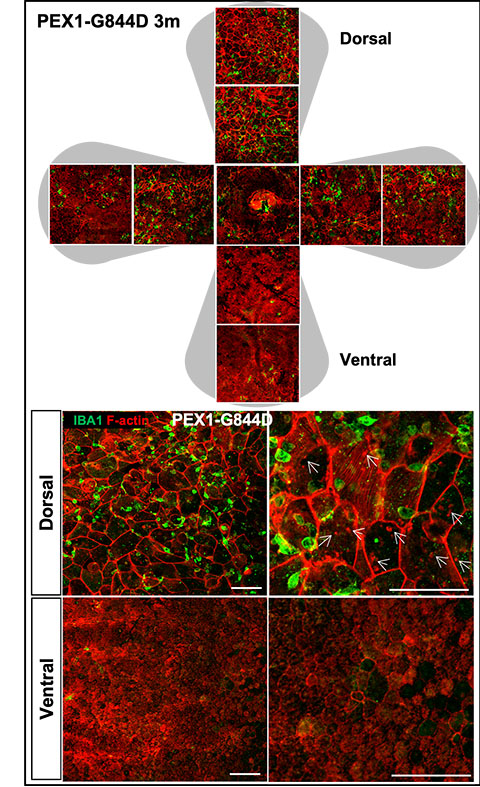
In a recent Journal of Lipid Research study, Samy Omri, a research associate in Nancy Braverman’s lab at McGill University Health Centre, and colleagues focused on mutations in the peroxisomal biogenesis factor 1, or PEX1, gene, the most common cause of ZSD. These mutations disrupt lipid biosynthesis and peroxisome assembly, leading to progressive retinal degeneration — a major cause of childhood blindness in affected individuals.
“Currently, there is no curative treatment, so supportive management aims to alleviate symptoms and improve patients' quality of life,” Omri said. “This includes dietary modifications to address metabolic imbalances, supplementation with essential fatty acids to support neurological function.”
To model ZSD-related retinal degeneration, the researchers used mice with the Pex1-G844D mutation, which mirrors the common human variant.
“The team has been deeply engaged in studying and treating peroxisomal disorders, and this project, investigating the influence of lipid metabolism on retinal health, naturally evolved from that work,” Omri said. “My expertise in retinal physiopathology, along with my interest in early molecular changes preceding inflammation and tissue degeneration, aligns well with the lab's research focus.”
The team focused on retinal pigment epithelium, or RPE, analyzing morphological, inflammatory and lipid changes in a mouse model at one, three and six months of age. They found that RPE degeneration could be detected by three months and worsened with age.
Using mass spectrometry imaging, generated by Pierre Chaurand’s lab, the group identified 47 lipids in RPE that were altered appeared before any visible structural degeneration in the retina. This approach allowed them to visualize the spatial lipid distribution in the retina, providing insight into early disease mechanisms.
“While previous work has largely focused on systemic disease characterization and functional recovery, our research shifts the focus to find new biomarkers for retinal degeneration and a deeper mechanistic understanding of ZSD pathology,” Omri said.
Next, the team plans to validate the clinical relevance of these lipid signatures, a step toward developing future diagnostic tools or therapeutic targets.
They also discovered progressive subretinal macrophage accumulation in ZSD mice, revealing a previously unrecognized inflammatory pathway potentially driving retinal degeneration. Future studies will investigate these inflammatory signal pathways to identify anti-inflammatory therapies that could slow or prevent retinal degeneration.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

The molecular orchestra of memory
Calcium, calmodulin and calcium/calmodulin-dependent kinase II form a molecular axis that turns fleeting neural activity into lasting memories. New research shows how memories are stabilized, and possibly even protected or repaired.

