Plasma membrane is no barrier to free fatty acid
Our understanding of how long-chain fatty acids cross membranes is changing based on recent work by James Hamilton’s laboratory at Boston University and others. Their research shows that unlike nutrients such as glucose or amino acids, which require a transporter, fatty acids can diffuse spontaneously through protein-free lipid bilayers and cells’ plasma membranes.
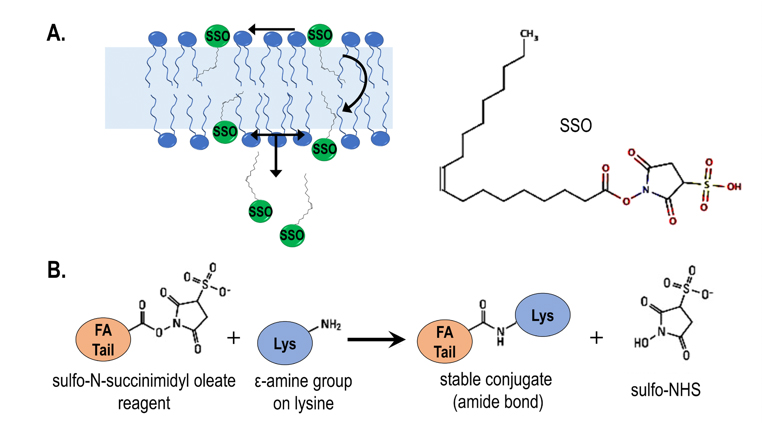
A recent paper in the Journal of Lipid Research by Anthony Jay and colleagues reveals how molecules previously thought to inhibit fatty acid transport specifically, including sulfosuccinimidyl oleate, or SSO, fit the diffusion model.
Control of fatty acid entry into cells is difficult to visualize. Researchers often have used fatty acid metabolites that become trapped in the cell to infer transport by plasma membrane proteins. The Hamilton lab has focused on distinguishing transmembrane movement from metabolism.
The lab made a seminal discovery that transport of fatty acids across a bilayer can be followed by pH changes. They combined a fluorescent fatty acid reporter with phospholipid vesicles enclosing a pH-probe to measure absorption in real time. They showed that natural fatty acids can acquire a proton near the outer leaflet, leading to equilibrium between neutral and ionized forms. The net neutral fatty acid spontaneously diffuses across the bilayer and then releases the proton near the inner leaflet, reducing the internal pH. This energy-free diffusion has been termed the “flip-flop” mechanism.
“Our studies also showed that all fatty acids studied … exhibit rapid flip-flop, and that this mechanism is reversible,” Hamilton said.
The researchers were excited by this finding but acknowledge that it does not exclude a role for proteins. Jay said, “If fatty acids can simply diffuse into cells, how could there be inhibitors of this transmembrane movement?”
The recent paper tests several proposed transport inhibitors, including the gold standard, SSO. Scientists had described SSO as a specific inhibitor of fatty acid entry into cells that acts on the fatty acid transporter CD36 without penetrating membranes. However, Hamilton’s team showed that SSO crosses membranes. Immunofluorescence using novel antibodies that bind SSO-linked fatty acids suggested that SSO modifies numerous proteins on the cell surface and interior, refuting the assumption of CD36 specificity.
The research informs nutritional considerations, Hamilton said. “Metabolism, and not the plasma membrane, controls the retention of fatty acids in cells, by trapping the fatty acids. Although membrane proteins may bind fatty acids or participate in metabolism, they cannot block diffusion in the surrounding bilayer.”
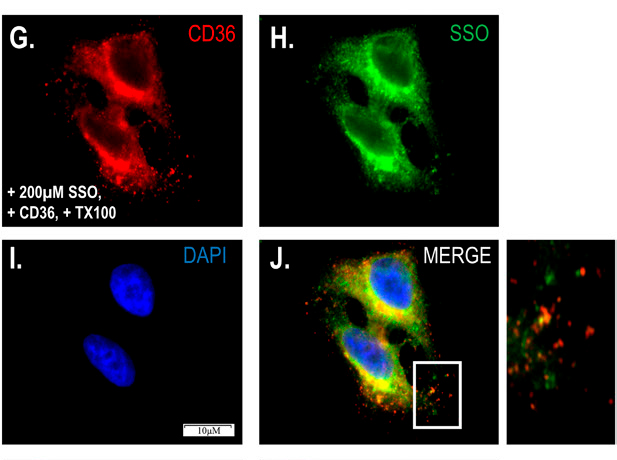
and the fatty acid transport inhibitor sulfosuccinimidyl oleate. The insert on the right suggest that SSO staining is not
exclusive to CD36.
In fact, cells with and without CD36 exhibited the same diffusion, but CD36 increased the content of intercellular lipids.
For nutrition, Hamilton emphasized, “Healthy fatty acids such as omega 3 fatty acids need to enter cells readily, whereas unhealthy fatty acids, such as trans fatty acids, cannot be excluded from cells and need to be reduced in the diet.”
Hamilton’s lab is moving on to clinical trials using a high concentration of beneficial fatty acids to improve stroke and heart attack outcomes. Meanwhile, he recommends taking omega-3 supplements and eating more nuts.
Another interpretation
According to Maastricht University researchers Jan Glatz and Joost Luiken, the Hamilton lab’s results also could be interpreted as a step toward integration of the argument that fatty acids diffuse across the cell membrane and the view that the protein CD36 is required for fatty acid uptake. In a Letter to the Editors of the JLR, Glatz and Luiken lay out their view that CD36 may be helpful but not necessary for lipid uptake.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Cracking cancer’s code through functional connections
A machine learning–derived protein cofunction network is transforming how scientists understand and uncover relationships between proteins in cancer.

Gaze into the proteomics crystal ball
The 15th International Symposium on Proteomics in the Life Sciences symposium will be held August 17–21 in Cambridge, Massachusetts.
Bacterial enzyme catalyzes body odor compound formation
Researchers identify a skin-resident Staphylococcus hominis dipeptidase involved in creating sulfur-containing secretions. Read more about this recent Journal of Biological Chemistry paper.

Neurobiology of stress and substance use
MOSAIC scholar and proud Latino, Bryan Cruz of Scripps Research Institute studies the neurochemical origins of PTSD-related alcohol use using a multidisciplinary approach.
Pesticide disrupts neuronal potentiation
New research reveals how deltamethrin may disrupt brain development by altering the protein cargo of brain-derived extracellular vesicles. Read more about this recent Molecular & Cellular Proteomics article.

A look into the rice glycoproteome
Researchers mapped posttranslational modifications in Oryza sativa, revealing hundreds of alterations tied to key plant processes. Read more about this recent Molecular & Cellular Proteomics paper.

