Examining mechanisms of protein complex at a basic cell biological level
A new study highlighting the importance of a large protein complex called the exocyst in cell growth, division and communication reveals new functions and mechanisms that are essential to how molecules move across a membrane through vesicles in a cell.

Understanding how these mechanisms work in normal cells at the basic biological level will inform future research into how those functions are disrupted in developmental and neurological disorders.
“It’s the first time a role for membrane fusion has been described for this complex and it’s a breakthrough in how we think about the way the exocyst complex works,” said Mary Munson, professor and vice chair for diversity in the Department of Biochemistry & Molecular Biotechnology, associate vice provost for equity in science in the Office of Health Equity and a co-corresponding author on the study published in Nature Structural & Molecular Biology. The research was done in collaboration with Tae-Young Yoon, professor of biological sciences at the School of Biological Sciences and Institute for Molecular Biology and Genetics at Seoul National University in Seoul, South Korea.
“Until our collaborative study, exocyst was understood to recognize and possibly tether secretory vesicles to the cell membrane, prior to exocytosis. In this study, we reveal biophysical studies that indicate exocyst playing several critical roles that directly facilitate fusion of the vesicles with the cell membrane to drive cargo delivery,” Munson said.
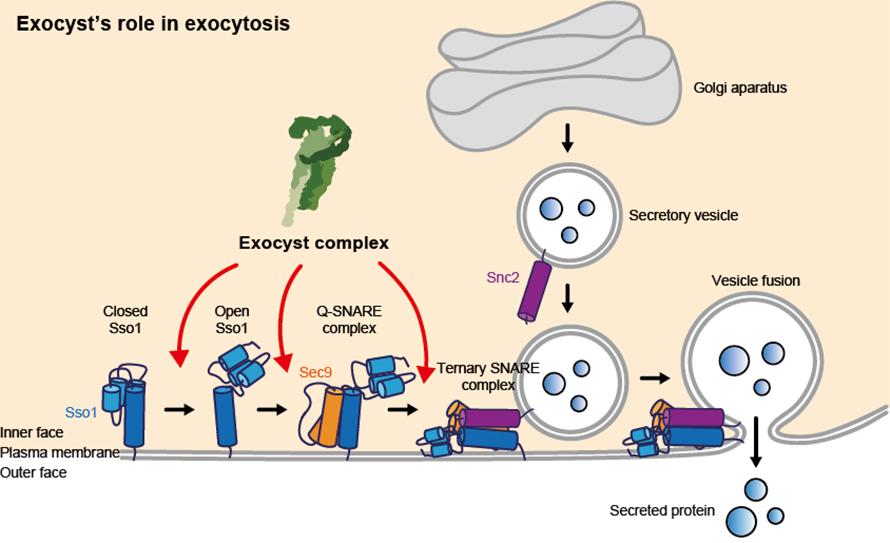
Robust control of exocytosis is critical for all cells to grow, divide and communicate properly. Dysfunction of exocyst regulation has been linked to many physiological problems in different organisms.
“It’s really an exciting development in our field, as the exocyst complex had not previously been shown to directly control vesicle fusion,” Munson said. “The complex recognizes the right vessel, the right place on the cell surface and the right time for the cargo to be delivered. It does that by talking to the membrane fusion protein.”
In 2023, Munson received a National Institutes of Health award for $3.3 million over five years to support this research.
Munson was recently named a 2024 recipient of a Zenith Award from the Association for Women in Science. The award honors senior career professionals with a lifetime of innovative achievements in STEM and a commitment to workplace diversity.
This article is republished from the UMass Chan Medical School News. Read the original here.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

