Should states keep samples from newborn heel blood tests?
Close to 4 million babies are born in the United States every year, and within their first 48 hours nearly all are pricked in the heel so their blood can be tested for dozens of life-threatening genetic and metabolic problems. The heel-stick test is considered such a crucial public health measure that states typically require it and parents aren’t asked for their permission before it’s done.
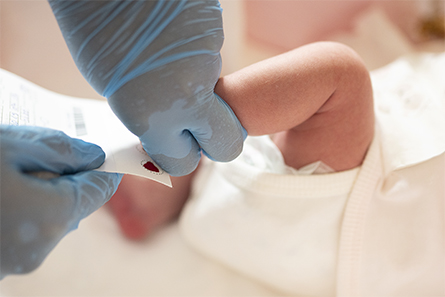
But the lab tests for newborn screenings generally don’t use all of the half-dozen or so drops of blood collected on filter paper cards. So states hold on to the leftover “dried blood spots,” as they’re called, often without parents’ knowledge or consent. In recent years, privacy-related concerns have grown about the sometimes decades-long storage and use of the material.
Some states allow the blood spots to be used in research studies, sometimes by third parties for a fee, or provided to law enforcement personnel investigating a crime. Permitting these or other uses without parents’ informed consent that they understand and agree to the use has prompted lawsuits from parents who want to make those decisions themselves and who seek to protect their children’s medical and genetic information.
In May, Michigan officials reportedly agreed to destroy more than 3 million blood spots as a partial settlement in a lawsuit brought by parents who said they didn’t receive enough clear information to provide informed consent for the blood to be used in research the state might conduct. The fate of millions of additional blood spots stored by the state will be determined at trial.
Philip L. Ellison, an attorney in Hemlock, Michigan, who is spearheading the suit, said he became aware of the issue when his son was born five years ago. Ellison’s son, Patton, spent his first days in the neonatal intensive care unit after his blood sugar levels dropped precipitously after birth. The next morning, Ellison said, he was approached by a hospital staffer who asked whether he wanted to sign a consent form allowing the blood from Patton’s heel-stick test to be donated for research.
“We don’t know what the future will bring in terms of information that can be extracted from our blood,” he said. How the rules for using that blood might evolve over time, he said, is difficult to know. “A program that first starts out for one purpose, to test for disease, has now crept into medical research and then to law enforcement,” he said.
Michigan is the rare state that asks parents for permission to use leftover newborn blood spots in research. Most do not, experts said. The state screens newborns for more than 50 diseases, such as cystic fibrosis and congenital hypothyroidism, because identifying and treating such illnesses early in a child’s life are crucial.
Afterward, whatever is left over is stored for up to 100 years and, if parents agree to it, may be used in research approved by the Michigan Department of Health and Human Services. Some recent studies have used de-identified blood spots to study the relationship between viral infection at birth and the development of autism later in life, as well as the impact of maternal exposure to manufactured chemicals known as PFAS on health outcomes.
Parents have also asked that their children’s blood spots be sent to researchers to help diagnose a disorder or to try to find a reason for a child’s death, said Chelsea Wuth, a spokesperson for the Michigan Department of Health and Human Services.
Michigan parents can request that the state destroy the leftover blood spots if they don’t want the state to hold on to them.
Since the 1960s, states have screened newborn blood for conditions that can lead to devastating physical or mental disabilities or death if they are not diagnosed and treated. The federal government recommends that roughly three dozen screening tests be performed, but some states conduct many more. Every year, an estimated 13,000 infants with serious medical conditions are identified through newborn screening programs, according to data published by the federal Centers for Disease Control and Prevention.
Many public health experts strongly support mandatory newborn screening as a critical component of infants’ clinical care. But some are receptive to giving parents a say in what happens to the blood after the screening.
“I have always believed that parents should be able to have the opportunity to say ‘yes’ or ‘no’” to having their newborns’ leftover blood used in research, said Dr. Beth Tarini, a pediatrician and the associate director of the Center for Translational Research at Children’s National Research Institute in Washington, D.C. “Since it is not part of the clinical care, it is a different standard of engagement with the parents.”
In Michigan, 64% of parents consented to participate, according to court documents in Ellison’s case.
Encouraging people to participate is important, some public health experts say, because the blood spot repositories provide a rare opportunity for population-level research. People of European descent are often overrepresented in genetic databases, which can skew the results of studies. But the newborn screening program includes virtually everyone born in the U.S.
“There’s strong evidence that research conducted on samples of white people creates disparities in the benefits of biomedical research for people who are not white,” said Dr. Kyle Brothers, a pediatrician and bioethicist at Norton Children’s Research Institute in Louisville, Kentucky.
After privacy-related lawsuits were brought in 2009 and 2011 by parents in Texas and Minnesota, respectively, millions of blood spots were destroyed.
Brothers said an unwillingness to participate in research programs reflects larger trends, including more emphasis on the individual and less on contributing to the general good.
To those who might argue that parents’ privacy concerns are overblown, a recent lawsuit in New Jersey raises troubling questions.
In a public records lawsuit, the New Jersey Office of the Public Defender and the New Jersey Monitor, a nonprofit news site, charge that the state police used a subpoena to obtain an infant blood spot of a child who is now 9 years old from the state’s newborn screening laboratory. The lawsuit says a DNA analysis was conducted on the blood spot so evidence could be gathered against the child’s father, who was being represented by the public defender’s office, in connection with a sexual assault committed in 1996. The effort allowed police to get the DNA information without having to show a court probable cause, the suit alleges.
The lawsuit seeks to find out how often in the past five years New Jersey law enforcement agencies have used the newborn screening lab as a tool in investigations and subjected defendants to “warrantless searches and seizures.”
New Jersey keeps the records on file for 23 years, said CJ Griffin, a lawyer representing the public defender’s office and the New Jersey Monitor in the lawsuit.
Griffin said her clients aren’t challenging the program to test newborn blood for diseases. “It’s more the lack of transparency, and safeguards, and information about storage, and we don’t have any information about appropriate use,” she said.
The New Jersey Department of Health doesn’t comment on pending litigation, spokesperson Nancy Kearney said. Kearney didn’t respond to a request for information about the state’s practices and policies related to the newborn screening program.
A recent Texas Law Review article found that more than a quarter of states lack policies on law enforcement access to newborn blood spot samples and related information and that nearly a third may allow access in certain circumstances.
In Michigan, the state gives law enforcement agencies dried blood spots only to identify the victim of a crime, Wuth said. “Typically, this means someone has been killed or gone missing,” she added.
Many clinicians and bioethicists say that standards for the use of blood spots need to be set.
“It’s nearly impossible for us to monitor the potential uses of our data,” said Andrew Crawford, senior policy counsel for the privacy and data project at the Center for Democracy and Technology. “That’s why need to put limitations on the use.”
This article was originally published on Kaiser Health News, a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Policy
Policy highlights or most popular articles

Lab coats and lounge chairs
ASBMB’s director of public affairs shares practical tips for talking about science outside the lab, from avoiding jargon to crafting an effective elevator pitch that builds curiosity and trust in scientific research.

And they’re off: Budget season begins in Congress
ASBMB’s director of public affairs explains the federal budget process after the president’s proposal, highlighting proposed cuts to science agencies, next steps in Congress and how ASBMB will advocate for research funding and mobilize members.

ASBMB members advocate for funding on Capitol Hill
In more than 120 meetings with lawmakers, 50 scientists called for sustained investment in NIH, NSF and DOE research programs.

Women’s health cannot leave rare diseases behind
A physician living with lymphangioleiomyomatosis and a basic scientist explain why patient-driven, trial-ready research is essential to turning momentum into meaningful progress.

Building a stronger future for research funding
Hear from Eric Gascho of the Coalition for Health Funding about federal public health investments, the value of collaboration and how scientists can help shape the future of research funding.

Councilors advocate for science on Capitol Hill
ASBMB Councilors meet with their elected officials to advocate for basic scientific research funding and training the next generation of scientists.

