JBC: How bacteria build efficient photosynthesis machines
Researchers facing a future world with a larger human population and more uncertain climate are looking to photosynthetic bacteria for engineering solutions to improve crop yields.
In the Journal of Biological Chemistry, a Canadian research team reports on how cyanobacteria finesse one of the most wasteful steps in photosynthesis. The study investigated the assembly of carboxysomes in which the bacteria concentrate carbon dioxide, boosting the efficiency of a critical enzyme called RuBisCO.
“Essentially everything we eat starts with RuBisCO,” said Matthew Kimber, a professor at the University of Guelph in Ontario and senior author on the paper.
The enzyme, which is made of 16 protein subunits, is essential for photosynthesis. Using energy captured from light, it incorporates carbon dioxide into organic molecules from which a plant then builds new sugar. Unfortunately, it’s not terribly efficient. Or, from Kimber’s point of view, “RuBisCO has a really thankless task.”
The enzyme evolved in an ancient world where carbon dioxide was common and oxygen was rare. As a result, it isn’t very picky in discriminating between the two gases. Now that the atmospheric tables have turned, RuBisCO often accidentally captures oxygen, generating a useless compound that the plant then has to recycle.
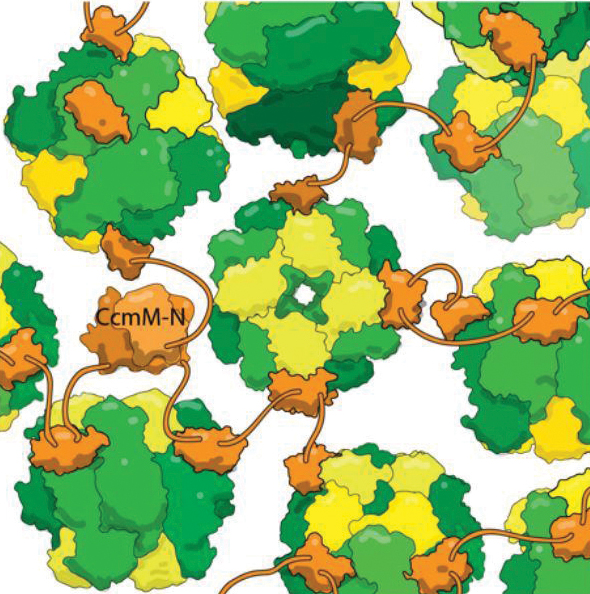
Cyanobacteria make few such mistakes, because bacteria collect their RuBisCO into dense bodies known as carboxysomes. The bacteria pump bicarbonate (simply hydrated CO2) into the cell; once it gets into the carboxysome, enzymes convert the bicarbonate into carbon dioxide. Because the carbon dioxide can’t escape through the protein shell surrounding the carboxysome, it builds up to high concentrations, helping RuBisCO avoid costly mistakes.
Kimber wants to understand the logic of carboxysomes’ organization. “They’re actually phenomenally intricate machines,” he said. “The cyanobacterium makes 11 or so normal-looking proteins, and these somehow organize themselves into this self-regulating mega-complex that can exceed the size of a small cell.”
One of carboxysomes’ most impressive tricks is self-assembly, which Kimber’s lab set out to understand. They looked at a protein, CcmM, that corrals RuBisCO enzymes into new carboxysomes. They knew that part of CcmM looks a lot like a subunit of RuBisCO — so much so that researchers suspect ancient cyanobacteria created CcmM by duplicating a RuBisCO gene.
Most scientists in the field believed that CcmM binds to the enzyme by usurping that RuBisCO subunit’s spot. But when Kimber’s lab took a detailed look at CcmM’s structure and binding, the results showed that was wrong. True, CcmM was similar in shape to the small RuBisCO subunit. But the complexes it formed still included all eight small subunits, meaning that instead of stealing a spot from a RuBisCO subunit, CcmM had to be binding elsewhere.
“This is very odd from a biological perspective, because if CcmM arose by duplicating the small subunit, it almost certainly originally bound in the same way,” Kimber said. “At some point, it must have evolved to prefer a new binding site.”
The researchers also found that a linker between binding domains in CcmM is short enough that “instead of wrapping around RuBisCO, it tethers (individual enzymes) together like beads on a string,” Kimber said. “With several such linkers binding each RuBisCO at random, it crosslinks everything into this big glob; you wrap a shell around it, and this then becomes the carboxysome.”
Scientists at another university reported last fall that they had succeeded in making tobacco plants with a stripped-down carboxysome in their chloroplasts. Those plants didn’t grow especially well, and the authors concluded that they had taken away too many components of the carboxysome; although it could be built in the chloroplast, it was a drag on the plants instead of a help.
A better understanding of how proteins like CcmM contribute to carboxysome construction and function could help bioengineers leverage carboxysome efficiency in the next generation of engineered plants.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

