UTSW researchers discover how food-poisoning bacteria infect the intestines
Researchers at the University of Texas Southwestern Medical Center have discovered how a bacterium that infects people after they eat raw or undercooked shellfish creates syringe-like structures to inject its toxins into intestinal cells. The findings, published in Nature Communications, could lead to new ways to treat food poisoning caused by Vibrio parahaemolyticus.

“We have provided the first visual evidence of how a gut bacterial pathogen uses this assembly method to build a syringe to deliver a lethal injection to intestinal cells,” said Kim Orth, a professor of molecular biology and biochemistry and a Howard Hughes Medical Institute investigator at UTSW. “This work provides a new view of how enteric bacteria when exposed to bile acids efficiently respond and build a virulence system.”
V. parahaemolyticus, commonly found in warm coastal waters, is a leading cause of seafood-related food poisoning. People infected often have diarrhea, cramping, vomiting, fever and chills.
Researchers knew that V. parahaemolyticus injects molecules into human cells using a structure called the type III secretion system 2 (T3SS2). However, these syringes, composed of 19 different proteins, are not produced or assembled until the bacteria are inside the intestines. Scientists were not sure exactly how this occurs.
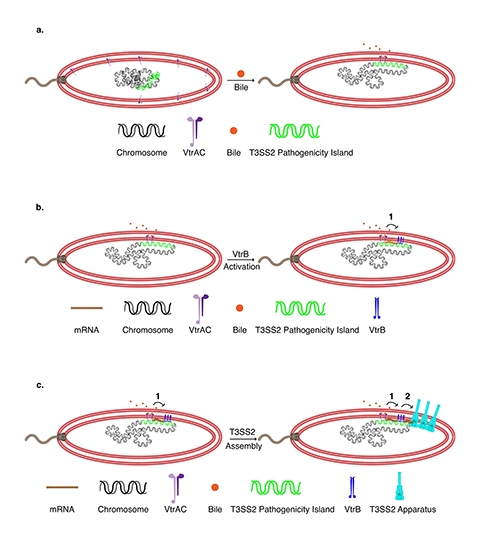
The latest findings build on the work of a previous study by the Orth lab. Orth and her colleagues tagged components of the V. parahaemolyticus T3SS2 with fluorescent markers and used super-resolution microscopy to track their locations as the bacteria were grown in different conditions. The researchers discovered that when V. parahaemolyticus is exposed to bile acids — digestive molecules in the intestines — the bacteria move DNA containing the T3SS2 genes close to their membrane.
Then, at the exact site where the T3SS2 is needed, V. parahaemolyticus transcribes that DNA into RNA, translates the RNA into protein, and assembles the components of the T3SS2 through the membrane in a process known as transertion. “It is like watching the assembly of a factory that produces a large molecular machine within an hour,” Orth said.

These steps were previously thought to occur in more disparate locations around a cell, but pulling the machinery together into one place on the bacterium’s membrane likely helps V. parahaemolyticus more quickly and efficiently build the T3SS2 and infect cells. Since other disease-causing gut bacteria contain molecular components similar to V. parahaemolyticus, the phenomenon of transertion may be widely used, the researchers hypothesize.
“Our findings imply that other gastrointestinal pathogens may also use this mechanism to mediate efficient assembly of complex molecular machines in response to environmental cues,” said UTSW research specialist Karan Kaval, first author of the paper.
More work is needed to know which bacteria use transertion to build their T3SS structures and whether drugs could be developed that block transertion to treat V. parahaemolyticus infections.
UTSW researcher Jananee Jaishankar also contributed to this study.
This article was first published by the University of Texas Southwestern Medical Center. Read the original.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

The molecular orchestra of memory
Calcium, calmodulin and calcium/calmodulin-dependent kinase II form a molecular axis that turns fleeting neural activity into lasting memories. New research shows how memories are stabilized, and possibly even protected or repaired.

Differences in pili structure modulate bacterial behavior
Researchers demonstrate how small changes in the structure of hair-like protein appendages can affect the behavior of Acinetobacter bacteria.

Cholesterol regulatory genes predict liver transplant outcomes
Researchers identify a link between cholesterol-regulating genes and liver transplant success, which could improve donor screening and patient outcomes.

