From the journals: JLR
We offer a selection of papers on a variety of topics recently published in the Journal of Lipid Research.
Chemical activator of GPR120 affects fat taste detection
In addition to sweet, sour, salty, bitter and umami, there might be a sixth taste: fat. Researchers have identified two receptor proteins, CD36 and GPR120, that are involved in detecting fat taste in both humans and mice. Mice that don’t express any GPR120 protein, for example, have less preference for fatty acids than normal mice.
Studies also have shown an association between fat taste sensitivity and obesity. In both mice and humans, GPR120 deficiency led to increased obesity. Thus, obesity might be modulated by tricking the body into thinking it is consuming fat when it’s taking in a small molecule that binds a fat taste receptor.
In a recent paper in the Journal of Lipid Research, Babar Murtaza from the University of Burgundy in Dijon and colleagues report that they tested whether a small molecule that binds GPR120 could be used for such a treatment. TUG891 previously had been shown to reduce obesity in mice.
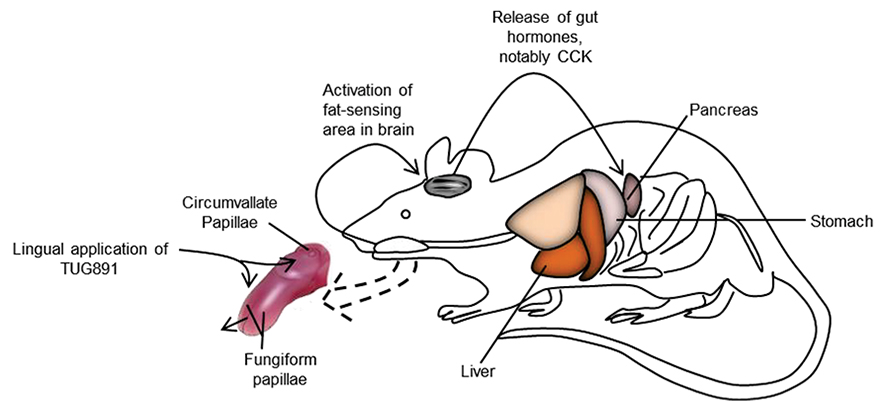
In studies using mouse and human taste bud cells, the researchers found that adding TUG891 or linoleic acid, a fatty acid, could promote calcium signaling through GPR120. They also tested whether TUG891 could activate the tongue–brain–gut loop by applying TUG891 or linoleic acid directly to the tongues of sedated animals. In both cases, pancreatic and bile duct secretions increased, as did blood circulation of the gut hormone cholecystokinin. The treatment also had an anti-inflammatory effect: The concentration of adiponectin, an anti-inflammatory cytokine secreted by fat tissue, in the blood increased, while levels of inflammatory cytokines decreased.
In behavioral tests, the researchers found that mice typically preferred drinking a solution of water, linoleic acid and a stabilizer over a solution of water and the stabilizer alone. Adding TUG891 to the linoleic acid solution removed any preference. These findings suggest that TUG891 may be helpful in human treatments to decrease consumption of fats and reduce obesity.
Breast cancer cells acquire lipids by endocytosis
Researchers long have thought that breast cancer tumor cells synthesize lipids as they multiply; more recent studies show that cancer cells also can take up lipids. In a recent paper published in the Journal of Lipid Research, Leslie Lupien of the Norris Cotton Cancer Center and researchers in the U.S. and Belgium identified a new pathway for lipid uptake. Experiments in multiple breast cancer cell lines showed that very low-density lipoprotein, or VLDL, is taken up via receptor-mediated endocytosis through interactions with surface-bound lipoprotein lipase, or LPL, and the VLDL receptor. The researchers propose that LPL may be binding free VLDL and concentrating it at the cell surface, thereby promoting VLDL uptake by the VLDL receptor. These findings have therapeutic implications and add to an increasing body of evidence that targeting fatty acid synthesis alone may not be effective in certain tumor types.
Triple therapy to treat atherosclerosis
Atherosclerosis is the leading cause of cardiovascular disease. Statins can help lower the low-density lipoprotein cholesterol that causes plaque, but treatment typically leads to only a modest reduction in plaque volume. In a recent paper in the Journal of Lipid Research, Marianne Pouwer of the Netherlands Organization of Applied Scientific Research in Leiden and a team of researchers in the U.S. and the Netherlands identified a combination of drugs that not only slowed plaque progression but also reversed it and improved plaque composition.
The researchers fed mice a high-cholesterol, high-fat diet and treated them with atorvastatin in combination with monoclonal antibodies alirocumab and/or evinacumab. The mice treated with the statin and one antibody had lower cholesterol and slower plaque progression compared to mice that received no treatment. Mice treated with a combination of all three drugs had plaques that were not only smaller but had less macrophage content and were less likely to rupture. These findings indicate that high-intensity treatment with multiple drugs could be a promising approach in humans as well.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

