World Tuberculosis Day
On March 24, 1882, microbiologist Robert Koch announced to colleagues at the Physiological Society of Berlin that he had found the bacterium that causes tuberculosis. Thanks to his work, and the work of many other researchers, the disease is now both preventable and curable. However, millions of people are still infected each year worldwide.
Along with the Centers for Disease Control and Prevention and other organizations around the world, the ASBMB is observing Tuberculosis Day 2018 by sharing research news from scientists expanding our understanding of Mycobacterium tuberculosis and a variety of perspectives on how the pathogen continues to affect our world.
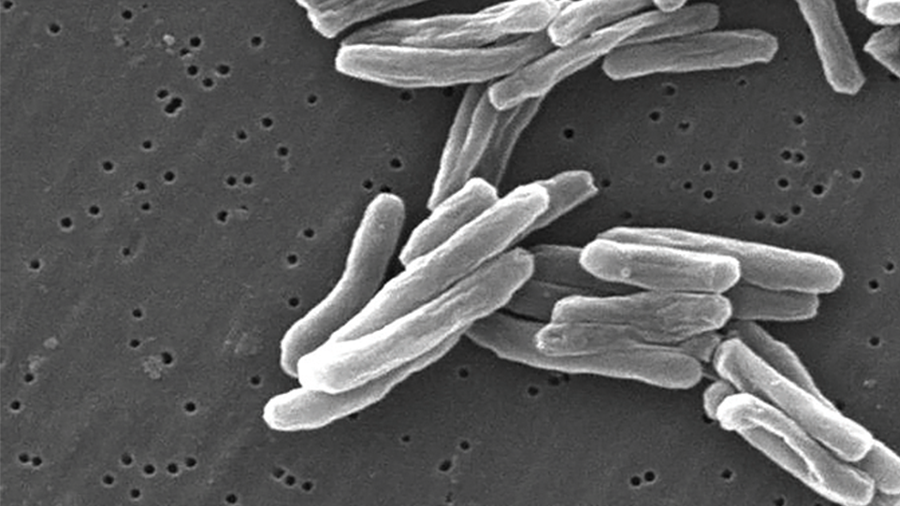 Molecular & Cellular Proteomics
Molecular & Cellular Proteomics
Improving TB vaccination
Mycobacterium vaccae, a species related to M. tuberculosis, is an immunogen of increasing interest as a possible vaccine. Scientists at the Chinese Academy of Medical Sciences used a proteomic analysis to pinpoint several immunogenic proteins in M. vaccae and reported their findings here.
A drug discovery resource
The first serine/threonine protein kinase interaction map for M tuberculosis was reported in MCP in June. The database, a resource for drug development, can be accessed here.
Bacterial immune evasion
In this study, researchers from Shanghai Jiao Tong University systematically assayed interacting proteins from M. tuberculosis and human proteomes, discovering a novel effector that binds to and suppresses a human ribosomal protein. Read more on their findings here.
Journal of Biological Chemistry
A prodrug-activating enzyme
TP053 is a promising anti-tuberculosis drug with potential to kill both replicating and nonreplicating bacteria. TP053 is a prodrug that must be activated by a redox enzyme from M. tuberculosis called DsbA. Researchers from across Europe characterized the role of DsbA in protecting the bacteria from oxidative stress. Read the article here.
Bacterial enzymology
The M. tuberculosis cytochrome P450 enzyme CYP121 is crucial for viability. But unlike the average cytochrome P450, CYP121 catalyzes a crosslink between carbon atoms. Researchers at the University of Texas at San Antonio used enzyme kinetics to characterize the reaction and propose a mechanism in this paper in JBC.
Hypoxic survival mechanisms
M. tuberculosis can survive in hypoxic microenvironments within the host alveolar macrophage by stealing and metabolizing host lipids. In a JBC article, scientists from India’s National Institute of Immunology identified an enzyme with lipase and protease activities key to hydrolyzing host lipids. Read the article here.
A function for a bacterial enzyme
After M. tuberculosis infects the host macrophage, it rapidly increases expression of the gene Rv2633, whose function was unknown. A research team at the University of Central Florida reported that the gene product is a catalase enzyme that protects the bacterium from oxidative stress during the host immune response in this paper in JBC.
Dual roles for a protein kinase
Mycobacterium tuberculosis virulence factor protein kinase G, or PKnG, prevents fusion of the host phagosome and lysosome, protecting the bacteria within the macrophage. Scientists from India’s National Institute of Immunology and the Indian Institute of Technology found that PKnG is also critical for bacterial survival during latency-like conditions, affecting the level of various substrates involved in metabolism. Read their article here.
Crystallizing a drug target
Antigen 85, which is important for the synthesis of the M. tuberculosis mycomembrane, is a target for a new class of cyclipostin and cyclophostin analogs. In a recent JBC article, researchers at the Centre National de la Recherche Scientifique in France reported the crystal structure of a cyclopostin analog bound to antigen 85, both demonstrating how the drug in question works and suggesting future approaches for drug design. Read their article here.
Predicting when rifampicin will fail
The antibiotic rifampicin is a first-line therapy for TB. In a recent paper, scientists from the Council of Scientific and Industrial Research in India identified a xenobiotic nuclear receptor from patients with rifampicin-susceptible infections who were not responding to therapy; the receptor dulls rifampicin’s effects. The finding suggests that adding a receptor antagonist may make a more potent therapeutic cocktail. Read more here.
Targeting the latent stage
The ribonucleolytic enzyme MazF is a promising drug target for the latent stage of tuberculosis. Researchers from Seoul National University in Korea described the protein’s structure in a paper accessible here.
Journal of Lipid Research
Improving diagnostics
Historically, diagnosis of tuberculosis has been challenging and subjective. In this article, scientists at the National University of Singapore used a phage display system to develop four antibodies specific for mycolic acid from the mycobacterial cell wall. While recent advances in nucleic acid diagnostics such as the GenXpert mean a lipid-based diagnostic is now less of a priority for tuberculosis, the approach could be used to develop diagnostic assays based on a variety of lipid biomarkers from different pathogens. Find out how the phage display system works here.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Catching tau in the act
Using a new proximity-labeling approach, researchers reveal how tangles of the brain-associated protein tau may disrupt RNA biology long before neurons die.
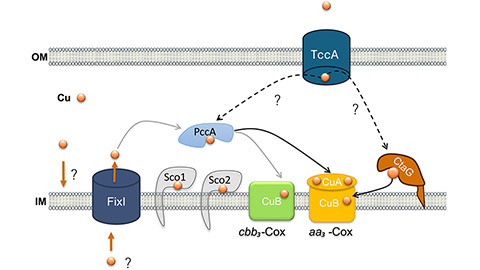
How copper delivery fuels bacterial respiration
Researchers identify the roles of several proteins in copper homeostasis in the aerobic bacterium Caulobacter vibrioides.
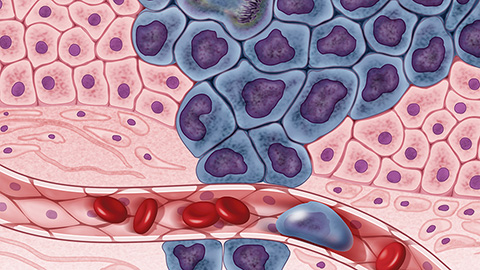
Revealing the glycoproteome of a cancer subtype
Researchers mapped the glycoproteome of extrahepatic cholangiocarcinoma and compared it to intrahepatic tumors. Differences in sugar modifications and immune cell content suggest new biomarkers and guide development of targeted immunotherapies.

Uncovering the mechanisms of a glycosylation disorder
Mutations in OGT, an enzyme that adds sugars to proteins, cause a rare neurological disorder. Using proteomics, researchers reveal how OGT interactions with TET proteins may trigger epigenetic changes and early neural defects.

Heat shock proteins as a promising breast cancer therapeutic
Researchers unveiled isoform-specific targets on heat shock protein 90 which may be beneficial in therapeutic development.

Optimized proteomic analysis of preserved biological tissue samples
Researchers have developed an optimized workflow for analyzing formalin-fixed paraffin-embedded tissue. This workflow provides an enhanced collection of unique proteins and phosphorylation sites for more detailed analysis of biological samples.
