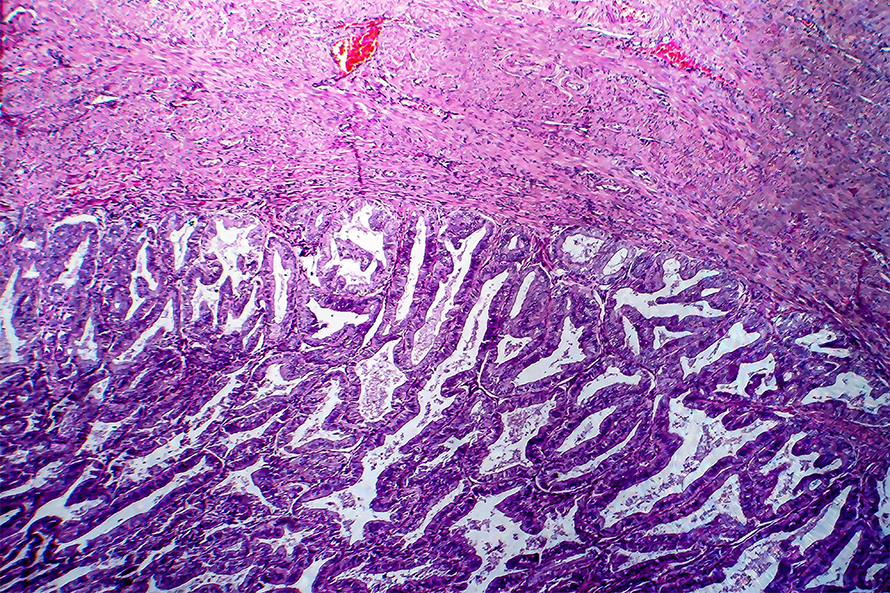
Researchers find potential new target for endometrial cancers
Researchers at Fox Chase Cancer Center have discovered a potential new target in the treatment of endometrial carcinoma' The research was published recently in Molecular & Cellular Proteomics.
Using multiplexed inhibitor beads and mass spectrometry, James S. Duncan, associate professor in the cancer biology research program at Fox Chase, and colleagues profiled the kinome of endometrial tumors and normal endometrial tissues and identified a network of kinases that were overexpressed, including serine/arginine-rich splicing factor kinase 1, or SRPK1.
According to Duncan, endometrial tumors frequently have alterations in protein kinases, a family of about 535 enzymes collectively termed the kinome. Kinases are altered in about one-quarter of all cancers and are considered highly susceptible to treatment with drugs because of their catalytic activity. However, only a small fraction of the kinome has been explored therapeutically.
Multiplexed inhibitor beads and mass spectrometry is a chemical proteomic strategy that allows researchers to simultaneously look at kinase levels in cells and tumors.
"From a cancer standpoint we can identify protein kinases that are up- or downregulated in cancer," Duncan said. "Ones that are upregulated are of interest because protein kinases play a role in cancer growth, survival, and metastasis, so they often represent potential targets."
The analysis showed that SRPK1 was overexpressed in endometrial cancer tissues and that this overexpression was associated with poor survival, suggesting that SRPK1 could be involved in key tumor-associated properties, Duncan said.
"We also discovered that in combination with therapy targeting growth factors—in this case EGFR—targeting this kinase produced strong drug synergy to kill these tumors," Duncan said. This synergy was found for endometrioid cell lines and uterine serous cancer cell lines, a disease subtype with poor outcomes.
To further explore the role of SRPK1, Duncan and colleagues want to apply proteomic technology to try to understand more about how SRPK1 works and conduct experiments to see if EGFR and SRPK1 inhibition has an effect on endometrial cancer tumor models.
"This project really sums up the concept of a lot of the work that Fox Chase is trying to do when it comes to research," Duncan said. "Peggy's Pathway selected our project for funding because they thought our approach of looking at kinase signaling looked interesting, and, as a result, we have found something that may be actionable."
Peggy's Pathway for Women's Cancer Care is a charity started in honor of Peggy Pettinato, who passed away from serous endometrial carcinoma. The organization's mission is to raise funds for research into innovative treatments and early detection of endometrial cancer, the most common gynecologic malignancy in the United States.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

