
Is chronochemotherapy coming?
Between a congressional effort to end daylight savings time, a New York Times feature on timed drug delivery, and reviews of circadian biology in several major journals, circadian medicine has had a buzzy year. The excitement is particularly high among oncology researchers.
At various intervals since the 1970s, long before the molecular gears of the circadian clock were understood, timed delivery of cancer medication has seemed to be just over the horizon. In 1979, when a group of oncologists published the paper “On methods for testing and achieving cancer chronotherapy,” cancer was the second most common cause of death in America, following heart disease. In 2020, the same was true — but cancer was catching up. In the intervening 40 years, scientists have learned a huge amount about how cancer works and how to treat it. Some cancers that once would have been a death sentence are now curable. Others have remained intractable despite decades of study and billions of dollars of research investment.
Now, with a better understanding of the molecular underpinnings of humans’ circadian clocks and more sophisticated measurements of physiological phenomena, researchers are taking a fresh run at the idea of circadian cancer therapy.
“There is an immense excitement (in the field),” said Francis Lévi, a French oncologist who has worked for decades on basic mechanisms and clinical studies of chronochemotherapy. He and his colleagues say they have begun to observe that drugs can be more effective, or cause fewer side effects, if delivered at certain times of day.
But others point to the long and disappointing history of circadian medicine as a reason to be cautious about the hype. The clock, said biochemist Aziz Sancar, “interfaces with everything. It’s logical to think that it should have an effect … but you have to demonstrate it experimentally.”
If treating cancer better were as simple as using the same drug at a different time of day, Sancar argues, wouldn’t we know that by now?
Daily rhythms of disease
Anyone who has awoken at 4 a.m. after a long-distance trip knows that the circadian cycle is a powerful regulator of human behavior. Its rhythms also can regulate disease. People with rheumatoid arthritis feel the most pain in the morning, while gout and asthma attacks are more than twice as likely at night. People with Alzheimer’s disease tend to become upset and uneasy around sundown.
Responses to external stimuli change by time of day too. In a famous experiment from the 1960s, mice injected at noon with lipopolysaccharide, a bacterial toxin mimicking sepsis, died 80% of the time; those injected at midnight died just 20% of the time. Conversely, some cholesterol drugs are best taken at night, when cholesterol synthesis is highest.

protein’s 3-dimensional structure.
Clock disruption and carcinogenesis
Does circadian disruption cause cancer? In humans, the answer has seemed to be yes. But preclinical models have yielded confusing results. Read more
Disruption of circadian rhythms has been linked to diseases, especially cancer. In epidemiological studies of shift workers, disruption to clock functions has been linked sometimes, but not always, with higher cancer rates (see “Clock disruption and carcinogenesis”). Meanwhile, studies on a variety of cancer biopsies indicate that some retain a daily cycle in clock proteins and proliferate in daily waves, but their timing relative to the rest of the body can vary.
In a recent study of breast cancer metastasis in the journal Nature, researchers at several hospitals in Switzerland reported that blood samples taken at 4 a.m., when patients usually would be asleep, contained many more circulating tumor cells than samples taken at 10 a.m. Cells from each time point also showed different growth profiles. When grafted into mice, the nighttime cells grew much more aggressively, suggesting that, independent of number, they might be better able to seed metastases.
Using transcriptomics, the researchers showed that circulating tumor cells captured at night expressed many genes related to mitosis, while those collected by day devoted more energy to translation and ribosome biogenesis. Oddly, however, the tumor cells also seemed to have no cellular time keeper; they had lost oscillation of their clock proteins.
How the cellular clock runs
The molecules that run the human circadian clock are expressed in almost every cell. The first described was a protein called CLOCK, short for “circadian locomotor output cycles kaput.” CLOCK works with binding partner BMAL1 as a transcription factor. When the two bind unfettered to DNA, they drive transcription of numerous genes. But that free access is possible for only part of the day. Most of the time, BMAL and CLOCK have additional binding partners that disrupt their ability to bind to DNA.
Their window of opportunity peaks a little after midday. As evening nears, their transcriptional activity produces a group of inhibitor proteins called cryptochromes, or CRYs, and period proteins, or PERs. When a cryptochrome binds the CLOCK–BMAL dimer, it recruits a pile-on of other proteins that becomes a powerful repressive complex.
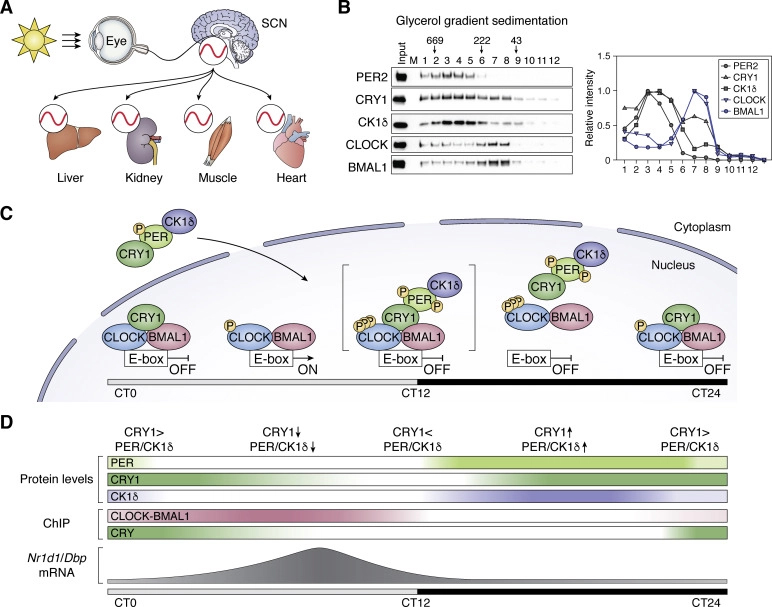
One kinase in the complex gradually phosphorylates CLOCK. Carrie Partch, a structural biologist at the University of California, Santa Cruz, who works on the assembly and diversity of circadian protein complexes, said that the enzyme’s inefficiency is a feature, not a bug. “We think that clocks rely on poor enzymes to help add the delay that leads to a 24-hour (cycle).”
By nightfall, CLOCK and BMAL are incapacitated. They relax their hold on the DNA. This turns off the faucet of CRY and PER protein production. As the night wears on, degradation reduces levels of CRYs and PERs, while phosphatases reverse CLOCK phosphorylation. As that happens, CLOCK and BMAL regain their ability to bind to DNA. One CRY, with especially high binding affinity, sticks around until dawn — at which point, it too begins to be degraded, eventually freeing CLOCK–BMAL to transcribe again.
Boiled down to its barest essentials, the cycle is simple. Night: off. Day: on. Repeat at roughly 24-hour intervals in almost every cell in the body. The central clock in the brain corrects any slippage.
The central clock
That central clock is composed of a cluster of neurons near the optic nerve, called the suprachiasmatic nucleus, or SCN, which receives light information directly from the retina, independent of the visual system.
Erik Herzog, a professor at Washington University in St. Louis, studies SCN function and communication. “In the same way we think about the atomic clock in Boulder, Colorado, sending out a pulse every morning at 3 a.m. … the SCN sends out daily signals to get everybody on local time,” Herzog said.
If a smartwatch were cut off from the atomic clock, it would continue to run but might drift out of accuracy. The same is true for cells: Each can oscillate on its own, but signals from the SCN keep them synchronized. Other parts of the brain turn pulses from the SCN into fluctuations in body temperature and circulating hormones melatonin and cortisol, each of which peaks at a signature time of day.
The SCN is a powerful pacesetter, but its control, researchers recently have learned, is not absolute. A short list of tissues, particularly in the skin and cornea, respond directly to light, even if the SCN has been rigged to run on a different schedule than an animal’s external environment. Also, in the course of human aging, central and peripheral clocks lose synchronicity.
Still, the SCN has an important coordinating role. For example, it is key to determining a person’s chronotype, or daily behavior pattern. Not every individual responds to light cues the same way; polymorphisms in clock genes, including the CRY and PER genes that mediate the CLOCK–BMAL pile-on, can determine how long an individual’s clock takes to complete a cycle — probably because they change the kinetics of protein complex formation, Partch said. Manipulating those genes in only the SCN can be enough to change an animal’s chronotype and turn a morning mouse to an evening mouse.
Whatever its inherent speed, as the cycle repeats itself in cells, it produces an ambient rhythm in gene expression. Nearly half of coding genes, and many noncoding RNAs, show circadian patterns in at least one tissue. Those transcripts affect the proteome, metabolome and cellular behaviors such as the cell cycle, DNA damage responses and apoptosis. Physiological functions such as sleep, body temperature, immune activity and feeding vary by time of day. Small wonder that researchers are asking whether medical interventions might also answer to the clock.
In discussions of drugs and the circadian rhythm, two types of interventions come up. A physician or a patient might try to adjust the clock through light therapy, timing cues such as exercise, or small molecules that interfere with the circadian oscillatory system. Or they might try to time medicines to reach their highest efficacy by working with biochemical and behavioral patterns that oscillate daily.
Drugging the clock
Scientists have found some evidence that bringing patients’ circadian rhythms into alignment using light and other cues can be beneficial. Timing food intake and exercise may help with Type 2 diabetes, and some (but not all) studies have concluded that cycling light conditions in a neonatal intensive care unit speeds development. But small molecules that alter the function of individual clock proteins have, so far, had little effect.
The clock is hard to drug, according to pharmacologist Tom Burris of the University of Florida, whose lab developed agonists for the clock protein called REV-ERB, a nuclear receptor that regulates BMAL1 expression. The drugs’ effects on mice are subtle; to see them, his lab had to dose the animals after they had been in the dark for several days. He thinks that’s because clock proteins are highly redundant, making the circadian cycle robust.
“I don’t think we can reset the entire rhythm, but we can modulate pieces of it,” Burris said. Because various clock proteins link to assorted other cell systems, he added, tweaking some of them might be useful, even if REV-ERB activation does not reset the sleep–wake cycle altogether.
Several groups have studied the effects of the REV-ERB agonists on tumor cells. Sometimes, their effects look promising. A lab at the Salk institute observed that they seemed to reduce autophagy and cause programmed cell death selectively in cancer cells. However, critics point out that the drugs also have effects in mice that lack the REV-ERB gene, suggesting that they may act through other targets.
Burris expects that, over time, “we’re going to understand more about how the oncogenic pathways are coupled to the circadian rhythm and be able to hijack some of the targets that are shared.”
of day. Researchers hope to find the optimal time to use drugs that target proliferation.
Still, because of this complexity, more physicians have focused on timing a drug for greatest efficacy. Jeffrey Field, a pharmacologist at the University of Pennsylvania, put it this way: “The evidence for drugging the clock is probably weaker than clocking the drugs.”
Clocking the drugs
An influential review article published in the journal Science in 2019 found that 75% of 106 trials testing the same drug at two times of day showed differences in either toxicity or efficacy. Roughly 20 of those trials tested cancer drugs. Drugs with a shorter half-life were more likely to show a time-of-day effect.
In a study that cited that work, researchers in the labs of Jeffrey Field and Penn colleague Amita Sehgal tested 126 drugs at six-hour intervals, comparing their effects on osteosarcoma cells that had a functioning circadian clock and others that did not. They found that the timekeeping cells were more sensitive to about half of the drugs at some times of day than at others. Focusing on a group of drugs that target the chaperone protein HSP90, which they previously had shown connects the clock with the cell cycle, they found that they were most effective if given just after the chaperone hit its lowest daily expression level.
“Proliferation was affected because a specific part of the cell cycle was regulated by the clock, and that part of the cell cycle is also targeted by many chemotherapeutics,” Sehgal, a circadian biologist, said.
In this case, timing the drug to match the circadian clock meant also timing it to match the cell cycle, which the clock can influence. Sehgal and Field proposed that links between the cell cycle and circadian rhythm may be among the most important reasons that some drugs show circadian effects. But other pathways also fluctuate with time of day: Researchers have suggested exploiting rhythms in DNA repair or inflammation, both of which interact with the clock, to get the most out of extant drugs. Unraveling causality in these complex interactions can be a challenge.
“Often I get the question, ‘Is the efficacy that you’re getting because you’re regulating the circadian system or some other piece?’” Burris said. “I’ve often said, you just can’t say that, because they’re connected.”
Besides hitting a target at the right time — which may mean that target concentration is high or low, depending on the drug — there’s a second mechanism by which dosing time might give a drug an advantage: It may cause lower toxicity at certain times of day. Most legacy chemotherapy drugs kill healthy and tumor cells somewhat indiscriminately, causing harmful side effects. Whether it’s killing more cancer cells or damaging other tissues less, improvement to a drug’s therapeutic index — the distance between an effective dose and a dose that causes harmful side effects — could be beneficial.
Finally, a drug may be metabolized differently over time. Besides the desired target and any collateral proteins that cause off-target effects, molecules that may cycle include the liver enzymes that convert a drug to its active form, the transporters that carry it into its target cell or across the blood–brain barrier, the enzymes required for degrading or secreting it, and other players that deliver an active pharmaceutical ingredient to the tissue where it acts.
Sehgal suggested that retrospective studies of people who happen to take a drug at different times are a good way to scan for effects without getting lost in mechanistic details. “Maybe that’s not the optimal time from the perspective of the daily rhythm in cortisol, but maybe it is from the point of view of when the target is up.”
Confusing clinical trials
Russell Van Gelder, a physician–scientist who chairs the ophthalmology department at the University of Washington, is skeptical that dosing at a specific time of day will yield dramatic medical advances. He said, “You’re taking extremely complicated physiologies that have multiple clock components running at the same time and assuming that there’s some phase that’s optimized and will have a very clear effect.”
He cited a history of clinical failures to show conclusive circadian effects. For example, a 2006 study of timed chemotherapy for patients with metastatic colorectal cancer found that timed dosing only helped some patients. Median survival was comparable between the study’s arms, but timed therapy reduced the risk of an earlier death in men by 25% and increased it by 38% in women.
No one is certain why women fared worse than men in this study. Author Francis Lévi, the oncologist in France, vehemently disputed the characterization of this study as a failure. Lévi’s group subsequently has conducted many studies defining the lowest-toxicity timing for various chemotherapies and continues to observe sexual dimorphism in optimal dosage times. He said, “It’s a complex issue, and this is why it has taken a long time and is not yet finished.”
But when a recent news article on circadian medicine cited the study but only mentioned the improved survival among men, Aziz Sancar, a biochemist at the University of North Carolina who shared the 2015 Nobel Prize for discovery of a DNA repair enzyme that helps to set the clock, was galvanized to speak up about his doubts about the field at large. Sancar’s lab had spent time looking for evidence that an optimal time exists when DNA-damaging chemotherapeutic agents might be most effective.
Doubled survival time among men, Sancar said, simply wasn’t what the 2006 study reported. “That got distorted, and all of a sudden people said chronotherapy cures cancer.”
Meanwhile, Sancar remained concerned about the half of study participants who had fared worse on timed treatment. “He felt that … this enthusiasm for timed chemotherapy could actually be harming people, or subgroups of people, and not helping them,” said longtime colleague Van Gelder.
In two review articles published back to back last year in the journal Science (with co-author Van Gelder) and the Journal of Biological Chemistry (with colleagues from UNC), Sancar laid out an argument that enthusiasm for chronochemotherapy exceeded the evidence that supports it.
Sancar’s argument is highly detailed but returns to several themes: He says that the chronochemotherapy literature glosses over negative data, overstates positive findings in reviews and generalizes inappropriately from small studies.
For example, he pointed to a widely cited 1985 paper reporting that a group of 31 women with ovarian cancer who received adriamycin in the morning and cisplatin in the evening experienced fewer side effects than those whose treatment schedule began in the evening; that study could not be replicated, Sancar said. In a study of thousands of patients with metastatic ovarian and uterine cancers, he added, the Gynecology Oncology Research Group “concluded that chronotherapy did not help.”
In an interview, Sancar also bemoaned widespread bias against publishing negative results, especially if they contradict previous findings. He said that he wants to see rigorous tests of the chronochemotherapy hypothesis considered objectively — including the disappointing studies.
Summing up their joint work, Van Gelder said, “It’s not that circadian rhythms don’t have anything to do with cancer. They have a great deal to do with cancer. It’s very clear that tumors disrupt rhythmicity, rhythmicity governs tumors, and there are deep mechanistic relationships between clock genes and oncogenes. However, the relationships don’t readily predict the appropriate timing for treatment.”
Presented with these critiques, Lévi wrote, “An ophthalmologist and a biochemist can hardly understand the methodologic constraints of clinical research and how much we learn from them to advance medicine irrespective of hypothesis validation.” One important finding from clinical studies, he said, has been that there is significant divergence between individual responses based on sex, lifestyle and genotype, meaning that drug timing probably will need to be personalized to be effective.
Lévi also cited a recent systematic review of chronochemotherapy studies that appeared in the Lancet Oncology. The review considered 18 clinical trials published from 1994 to 2021; Lévi was an author of six of them. Most investigated the effects of dosage timing on infused chemotherapeutic drugs, especially combinations including cisplatin and oxaliplatin. The study found that 11 of the 18 trials reported a reduction in toxicity, or harmful side effects, and none showed any reduction in efficacy. However, the review’s authors cautioned, it was impossible to isolate the effects of timing, since many of the studies also had altered the duration of infusions or the overall dosage patients received.
The case of temozolomide
“Historically, the research hasn’t been done carefully,” admitted Erik Herzog, the Washington University biologist. That makes it difficult for researchers to get support to test new circadian medicine hypotheses, such as the one he has been working on with WUSTL colleagues Joshua Rubin, a neuroscientist, and Jian Campian, a neurooncologist.
Herzog and Rubin teamed up to study timing in glioblastoma. They became interested in temozolomide, or TMZ, the best available treatment. The drug works by alkylating purine bases in DNA, causing double-strand breaks that halt replication. Patients take the pill daily for five days, take three weeks off to recover, and repeat indefinitely. TMZ has been a front-line treatment since 2005, but it’s no panacea; a glioblastoma patient can expect to live on average 16 months after diagnosis.
Emily Slat, a student in Herzog’s lab, found that cultured glioblastoma cells from both mice and humans keep a daily rhythm, and the time when they receive TMZ matters. Treating tumor cells early in the day killed many more of them. Herzog said that the timed effect seems to depend on DNA repair, not cell proliferation.

effective eventually. An artistic rendering shows the CLOCK protein.
“If you give temozolomide at a time of day when the clock is revved up and driving the expression of repair enzymes, you may not have much success with the drug,” Herzog said. “In fact, there are times of day when it looks like temozolomide does almost nothing.”
When she saw the results, Campian was intrigued but cautious. “I didn’t want to pull off a large study, put in a lot of resources, and then it doesn’t work,” she said.
Many treatments, she knew, have looked promising in preclinical models, only to fail in patients. Besides, trials are expensive. A principal investigator must pay institutional review board fees, compensate research assistants to screen and enroll patients and keep records in order, and devote staff time to analyzing data. Even though the drug in question already was prescribed to nearly every patient at the clinic, it was difficult to secure pharmaceutical companies’ interest without patent protection. When Campian wrote grants to the National Cancer Institute and several foundations, those groups also did not bite.
Instead, the team found an experiment already underway at their clinic. Many physicians, Campian said, recommend that patients take TMZ in the evening, when the nausea and fatigue it causes may be easier to bear. Other doctors recommend that it be taken in the morning.
WUSTL had a diverse enough group of prescribers for a retrospective study, which was easier to arrange since it would not impact clinical decisions. Among 166 patients who had passed through the clinic and whose dosing time could be determined, the study team found no difference in survival time between groups. However, among patients with methylation of the gene for DNA repair enzyme MGMT — a biomarker that physicians already know sensitizes cells to TMZ treatment — they saw a dramatic benefit from morning dosing.
Campian and Herzog next planned a small feasibility study that the university supported. They would ask patients to take TMZ either in the morning or the evening and compare their outcomes. Contrary to received wisdom, they found that among thirty-five patients on different dosing schedules, no significant differences in adverse side effects arose. However, there was also no significant difference in survival.
“It disappointed us, for sure,” Herzog said. But, he added, the study was too small to detect a positive effect reliably, especially one that might apply only to patients with methylated MGMT. In addition, the team did not control for patients’ chronotypes, or for disruptions to circadian rhythm that the cancer itself can cause. Continuing the study, the team has begun to use wearable activity monitors to determine how a patient’s internal clock compares to the wall clock.
To determine for sure whether there is any difference between taking TMZ in the morning and the evening, doctors would need to recruit several hundred patients, Campian said. She’d like to run such a study, because to her it seems more promising to get the greatest possible efficacy out of a drug that already has been shown to be effective and safe — safe, at least, as chemotherapeutic agents go — than to run the risks of developing a new treatment. She said that she is disappointed that funders do not seem to agree.
“It’s going to be a long time before (chronochemotherapy) becomes the norm, primarily because of the expense,” Herzog said. He added that studies that start by using cultured cells from patients to identify the times of greater drug efficacy for an individual may be a more cost-effective way forward.
Herzog said that most physicians and grant reviewers expect timing to have at best a mild effect on how well medicine works. He predicts that if robust evidence on one drug could be collected, it would help make the case for circadian therapies. But without much industry interest, it isn’t clear how such a blockbuster finding could happen.
“Once a drug company would show something, the others will too,” Lévi said. But he thinks that the industry as a whole is conservative and that no company is likely to move first on a concept that has failed in the past.
From chemotherapy to immunotherapy
At a recent American Society for Clinical Oncology meeting, Lévi counted five abstracts showing circadian effects of various drugs in different cancers. “I think the excitement about timing, and the magnitude of the benefit, is now an important fact,” he said.
Many of those drugs were antibodies, some of which activate the immune system. In contrast with broad-spectrum DNA-disrupting agents, these are biological drugs with single specific targets and may lend themselves to cleaner mechanistic hypotheses, Van Gelder said. Although they have very long half-lives, which blunts the effects of dose timing in small molecules, some antibodies have shown significant time-of-day effects, a puzzle no one has explained yet.
Immunotherapies, which recruit quiescent immune cells, may benefit from the immune system’s strong inherent circadian rhythms. Checkpoint inhibitors, antibodies that block a mechanism cancer cells use to evade immunity, are an example; one analysis suggests they may work best when more T cells can infiltrate the tumor and worst when T cell infiltration is low.
Some practical questions about implementing chronochemotherapy are closer than ever to being answered. For example, if a drug works best at certain times of day, do clinicians need to figure out whether a patient has a certain chronotype, or whether their individual tumor is cycling, before moving forward with treatment? Clinical researchers can control for patient chronotypes during trials using wearable activity monitors. To determine whether a tumor is in phase with its host, researchers may culture a tumor biopsy and add a luciferase reporter of circadian gene expression; or they may isolate circulating tumor cells and use an algorithm to evaluate the transcriptome and determine its point in the circadian cycle.
“We need to determine who are the patients with good rhythms, who are the patients with poor rhythms, and determine whether chronotherapy will be more or less effective,” Lévi said. But as long as it remains difficult to finance studies of chronochemotherapy, he added, “We will not know. We will not advance.”
Despite the challenges of demonstrating a conclusive effect for chronochemotherapy, many researchers expect the approach to bear fruit eventually, especially as the tools for personalized medicine improve doctors’ ability to tailor treatment plans for individuals.
Still, Van Gelder said, “I think the odds that we’re going to find some drug that is completely ineffective at one circadian phase and phenomenally effective at another are fairly low.” After all, most drug targets that fluctuate in response to the clock change by at most a few fold. “They don’t go from zero to a thousand.”
Nonetheless, Sancar said, the prospect of curing cancer with circadian rhythms is an appealing one, even for him. “All scientists, all human beings want their work to be relevant.”
Clock disruption and carcinogenesis
Does circadian disruption cause cancer? In humans, the answer has seemed to be yes. But preclinical models have yielded confusing results, making it hard to say why people who do shift work develop cancer at higher rates in epidemiological studies.
“It’s been almost a two-decade-long conversation now,” said biologist Carrie Partch at the University of California, Santa Cruz. “I don’t think there’s a really clear answer … in terms of ‘a particular cancer has a particular type of effect on the clock,’” or a particular disruption of the clock promoting particular cancers.
In mouse models, she said, removing different clock genes from different genetic backgrounds has led to results that are difficult to compare. In cell lines, it can be tricky to choose an appropriate control for an oscillating tumor tissue.
According to Aziz Sancar at the University of North Carolina at Chapel Hill, publication bias — the tendency for positive results to get more attention than negative findings — makes it harder to evaluate the evidence. For example, he described a colleague who had difficulty publishing her observation that, contrary to a previous high-profile paper, mice lacking two Period genes do not show higher cancer rates than their unmodified relatives.
According to French oncologist Francis Lévi, there are at least eight types of cancer whose prognosis is significantly worse if circadian signaling is disrupted. But studies also have shown that cancer — and its associated stress and difficult medical treatments — can disrupt sleep, which may impair circadian rhythms. The complex causal relationships complicate not only studies of whether circadian disruption can cause cancer but also studies concerning chronochemotherapy.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.


