Alternatives to animals
When Thomas Hartung was a medical student in Germany in the early 1990s, he interviewed a 35-year-old mother of three who was waiting for a liver transplant. Without the transplant, she would die, and Hartung recalls thinking, “I would do any experiment on mice if I could save this woman.”
Animal experiments have been the foundation of most medical advances in the past century, including vaccination technologies, cancer treatments and new treatments for neurological diseases. According to a 2007 report, “Medical Advances and Animal Research,” most Nobel laureates in physiology or medicine used animals in their research, considering it unfortunate but necessary.
The ethical obligation of researchers to reflect on their use of animal models is outlined in a set of principles known as the three R’s, which most nations use as a guideline for laws governing animal experiments. Zoologists William Russell and Rex Burch introduced the three R’s — replacement, reduction and refinement — in “The principles of humane experimental technique”in 1959. The three R’s stipulate that researchers first determine whether their animal experiment can be replaced with a nonanimal experiment. If this is not possible, experimenters should strive to reduce the number of animals they use. Finally, the experiment should be refined to minimize animal suffering.
Hartung, who successfully replaced animal experiments with cell cultures in his graduate research, is now director of the Center for Alternatives to Animal Testing, or CAAT, at the Johns Hopkins Bloomberg School of Public Health. He believes the biggest step researchers can take toward reducing animal experimentation is a change in mindset; animal models have been useful for so long, it’s hard to believe that there are better models out there, even if the data exists.
Hartung suggests that scientists start with a commitment to reexamine the three R’s each time they design an experiment: “Ask yourself: Are there new technologies? Can I use fewer animals? If you strive for this, it is a good compromise. Be open to truth and willing to change your mind.”
Promising techniques
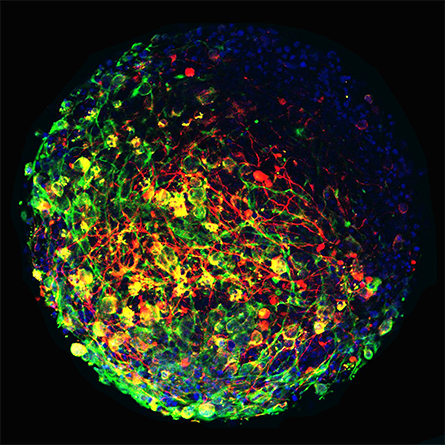
A growing number of techniques that can replace animal experiments have been developed in recent decades. In addition to being more humane than research on animals, these alternatives often produce results that predict human health outcomes equally well. Human cell lines, organoid systems and computational methods are the three most promising new technologies.

In the mid-20th century, the only cells scientists could grow successfully in a dish were derived from cancers. Cancer cells could be cultured easily because they grow and survive better than noncancer cells, but they were not appropriate for studying the physiology of a healthy cell. Eventually, scientists determined that stem cells — cells that are not fully developed — could be grown in a dish because they have an inherent ability to self-renew.
Initially, researchers could get stem cells only from embryos, an ethically complex and heavily scrutinized practice. Now, cells scraped from inside the cheek or from the skin of adults can be transformed back into their early developmental state. These induced pluripotent stem cells can be developed into any type of cell researchers want to study, making them a powerful research tool. These healthy human cells reflect the unique genetic background of the human from whom they were taken, allowing researchers to study the unique disease mechanisms of an individual.
When Hartung was a student researcher working with cell cultures, he said, he performed animal experiments just to prove to his colleagues that his cell system could predict human physiology as well as mouse models.
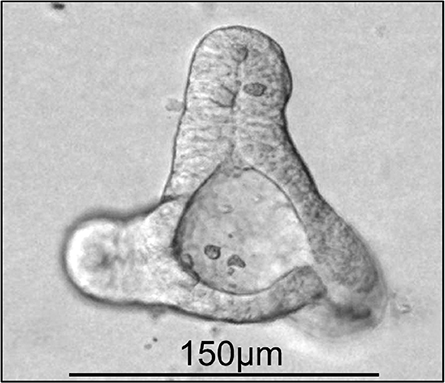
Although progress has been made in the development of better cell culture systems, a major justification for animal research is that cells in a dish cannot reflect completely what is happening in the human body. Cell cultures typically consist of one type of cell arranged in a flat layer in a Petri dish. In the body, not only are different types of cells interacting in any given organ, but these cells are organized into a complex 3D architecture. Organoid systems attempt to re-create this complex cell diversity and architecture outside the body.
Eva Rath, a postdoctoral researcher at the Technical University of Munich, studies the intestine using an organoid system called a “minigut” made from a small piece of intestine removed from a patient during a biopsy or bariatric surgery. Each minigut reflects the unique physiology of the patient. This is useful, as gastrointestinal disorders are complex. The same symptoms can have different causes from person to person because each individual has a unique immune system and collection of bacteria in their gut.

“It’s an individual disease,” Rath said.
Using miniguts, Rath and colleagues have discovered how inflammation can disrupt cellular metabolism and contribute to Crohn’s disease. Organoid systems have been developed to model human lungs, blood vessels, pancreas and even brains.
In addition to human cell culture systems and organoids, advancements in computational methods have made it possible to learn from computer models of disease. Before the advent of artificial intelligence, a computer was only as smart as the person who made it, and programs were only as smart as the person who coded them. According to Hartung, computational methods have made significant strides in the last decade.
“Now, computational methods can find patterns in data that humans never saw,” he said.
This is especially true in the area of computational toxicology, where algorithms that use similarities in chemical structure to predict toxicity outperform the reproducibility of animal tests, according to a 2018 report in the journal Toxicological Sciences. These new technologies make it possible to extrapolate existing data to gain new, useful insights without using animals.
Small but meaningful differences
In addition to saving animals, these new technologies often predict human health outcomes better than animal models because they use human cells or data. Although humans and the animals used as scientific models share a large percentage of their DNA, small genomic differences can have considerable effects.
Research on COVID-19 exemplifies this. Nicole Kleinstreuer, director of the National Toxicology Program’s Interagency Center for the Evaluation of Alternative Methods, or NICEATM, noted that mice don’t get severe COVID-19 the way humans do. “They catch it, but they don’t get sick,” Kleinstreuer said. “They don’t need to go on ventilators. They don’t die from it.”

Moreover, researchers have found that drugs tested in mice, the most common model organism, often fail clinical trials in humans and that preclinical studies using mice are often not reproducible. This could be because such studies are often statistically underpowered. Although it may seem counterintuitive, sometimes an individual experiment must use more animals to avoid wasting animal lives. If an experimenter uses only a few animals per condition and that is not enough to see the difference that they expect to see, they might get a false negative result. In such a case, the animals used in the experiment were wasted. They died, no biological insight was gained and the experimenter will perform underpowered animal experiments repeatedly. Ultimately, making sure that necessary animal experiments are appropriately powered is a way minimize animal use overall. Another reason mouse studies may fail clinical trials is that in many cases, even though humans and mice have similar genes, these genes serve different biological functions in animals and humans.
The current culture around alternatives to animal experimentation may prevent the timely implementation of new technologies. According to a 2019 report by David Lewis of Leeds University, there is low adherence among researchers to the three R’s guideline of replacing animal experiments when possible. “Despite the effort and resources being devoted globally to the development of alternatives, there is considerable reluctance amongst the research community to adopt them,” Lewis wrote.
In 1995, Hartung and a colleague found a way to test for the presence of fever-causing toxins in pharmaceuticals without using animals, forever changing the pharmaceutical testing industry. Typically, pharmaceuticals have been injected into rabbits to test for toxins. The researchers developed a method to test for toxins in donated human blood that could better predict adverse outcomes for humans. In their test, blood is mixed with the pharmacological compound. If toxins are present in the compound, the blood cells start to produce the immune protein interleukin-1-beta. By measuring the amount of this protein in the blood, scientists can determine whether a given compound contains toxins.
Hartung’s passion for alternatives to animal testing stems from his belief that overreliance on animal models hurts people. “If I say, ‘These poor rabbits,’ they say, ‘Why should I worry about them? It is so much more important to know what is toxic to humans,’” he said. “I say ‘No, it is so much more important to know what is toxic to humans, and this is why I don’t want to use rabbits.’”
Validation and policy
For scientists to reduce reliance on animal experiments, alternative methods must be available and validated. The availability of such alternatives varies widely among research areas. For example, it is much easier to get tissue for organoids from the intestine than to get it from the brain. A neuroscience researcher interested in studying a behavior needs an animal to model that behavior. Hartung understands that, for some scientists, animal experiments are the only appropriate tool to answer their research questions.
“If you have nothing better, I’m happy to endorse the animal experiment,” he said.
Even when an animal model is the best option, scientists often overestimate what that model is delivering. According to Hartung, this may be due to pressures from funding agencies and publishers. While researchers should be aware of the limitations of any animal model, this mindset doesn’t work in the quest for publication and grants — especially when reviewers are convinced that animals are the only way to ensure results are translatable.
“If you want to publish your findings in a cell culture model, most likely your reviewer will say ‘Where’s the animal experiment to validate this?’” Hartung said. “They don’t understand that human cell line systems can give results that are as good as or even better than the animal experiment. They say that without a mouse model, research done in human cells has no value.”

How can researchers be convinced to trust and use advancements in medical technology that do not use animals? Sonja von Aulock, editor-in-chief of the journal ALTEX, Alternatives to Animal Experimentation, believes that public policy could be key to getting researchers to consider nonanimal models.
“In Europe, animal testing for cosmetics was banned in 2013,” von Aulock said. “At that time, they didn’t have the alternative methods that they would need to test cosmetics without animals, but they innovated. Sometimes, you really need those higher-level decisions that say, ‘Come on, people. You’ve got to make this happen.’”
Minigut researcher Eva Rath agrees that policy plays a significant role in promoting alternatives and thinks science publishers should lead the charge. “Scientific journals should enforce their own criteria for accepting publications,” she said. “You should need to show that you use as few animals as possible.”
In her role at the NICEATM, part of the U.S. Department of Health and Human Services, Kleinstreuer connects the science of animal testing alternatives to policy development. Her office “conducts data analyses, workshops, independent validation studies, and other activities to assess new, revised, and alternative test methods and strategies,” according to its webpage.

her miniguts are growing to remove the waste and add new nutrients.
Results from these NICEATM studies provide support for the Interagency Coordinating Committee on the Validation of Alternative Methods — a committee representing 17 federal regulatory and research agencies including the departments of Agriculture and Defense, the Environmental Protection Agency, the Food and Drug Administration, and the National Institutes of Health. Through the NICEATM, representatives from these agencies ask scientists to design research to inform their policy decisions, and scientists communicate their needs to policymakers. For example, in 2018, analyses from the NICEATM informed the EPA’s decision to adopt a new policy to accept defined combinations of in vitro and computational methods instead of mouse tests when examining pesticide ingredients for skin sensitization.
Funding and revolution
Beyond creating programs to promote alternative research methods, policymakers also must allocate funding to those programs, Hartung said, adding, “Where there is funding, the science morphs and changes.”
Funding also is needed for the infrastructure required to support nonanimal models. “Labs have been working with animals forever,” von Aulock said. “Changing to other technology can be a big and expensive step.”
Kleinsteuer said the NIH is trying to expand the funding opportunities around human biology–based models.
It is a tall order to shift the collective mindset of scientists. Hartung draws on an idea from science philosopher Thomas Kuhn, saying, “Science doesn’t change continuously, but in waves. It takes a lot to believe that the current paradigm is not fitting anymore, accept something new, and suddenly change to a new paradigm. This is called a scientific revolution.
“Right now, we are entering a phase of revolution. Science is not giving justice to new methods because the old farts are reigning. These are the ones thinking that their methodologies are right and should continue to be used.”
Von Aulock agrees that the revolution starts with young scientists. “Question the status quo,” she said. “Think about whether you agree with how it has been done for the last 30 years. You are the generation that can make change. If you continue to plow on without being open to new developments, then things are not going to change.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

