Researchers investigate self-regulation of an enzyme with critical cellular functions
The lab of Kathy Gould at Vanderbilt University School of Medicine used a multidisciplinary approach that included structural biology, biochemistry and molecular biology to investigate the regulation of the CK1 enzyme family. The research was published in the journal Molecular Cell.

The work was led by postdoc Sierra Cullati and carried out in conjunction with research assistant professor Jun-Song Chen and scientists from Goethe University and the Structural Genomics Consortium in Frankfurt, Germany, and from Harvard University,
CK1 enzymes are a family of multifunctional kinases — enzymes that can phosphorylate, or add phosphate groups to, other proteins — that are critical for several cellular functions including DNA repair, endocytosis and mitotic checkpoint signaling. Regulation of CK1 enzymes is exceptionally important as dysfunction of these enzymes contributes to several conditions that include cancer, neurodegenerative diseases and sleep disorders.
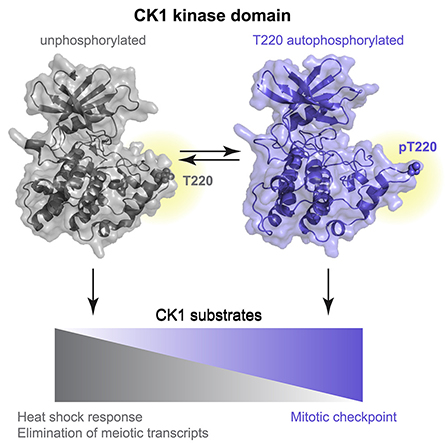
There are seven CK1 enzymes in mammals that perform different functions, but they are highly conserved in their catalytic domain, the region responsible for phosphorylation. Gould and colleagues found that one mechanism of CK1 activity, and thus one mechanism of regulation, is the self-phosphorylation of a conserved amino acid residue in its catalytic domain.
The researchers further investigated how this self-phosphorylation regulates activity and discovered that phosphorylation at this site altered the substrate specificity of CK1 enzymes. Substrate specificity refers to the determination of which other proteins the CK1 kinases will phosphorylate, which in turn determines which pathways within a cell get activated. In general, the phosphorylation state of CK1 enzymes controls their function — or dysfunction — within a cell. Determining which pathways are controlled by the phosphorylated versus non-phosphorylated states of the enzymes is a step toward the development of better treatments with fewer side effects for the diseases caused by enzyme dysfunction.
The Gould lab and collaborators hope to build upon this work by determining other sites of CK1 self-phosphorylation and investigating the pathways they regulate; there are several potential self-phosphorylation sites clustered together on one end of the protein, for example, that intrigue the researchers. Additionally, they plan to investigate how the discovered phosphorylation sites work together to provide additional control under different cellular conditions, such as cellular stress.
This article was republished with permission from the Vanderbilt School of Medicine. Read the original.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Glaucoma model links immune signaling to disease progression
Researchers at Duke University determine genetic variations that could increase the risk of developing glaucoma.

Uncovering the molecular roots of fatty liver disease
Physician–scientist Silvia Sookoian discusses her path from hepatitis C care to MASLD research, her use of multi-omics to study steatotic liver disease, and how lipid metabolism and genetics are reshaping understanding of MASH and liver health.

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

