Scientists sweep cellular neighborhoods where Zika hides out
Most people infected with Zika never show symptoms. But the virus sometimes causes severe disability — from microcephaly in babies to weakness or partial paralysis in adults — and there is no treatment. In a paper in the journal Molecular & Cellular Proteomics, researchers report a comprehensive study of how the virus interacts with host cells. One of their findings gives insight into how Zika escapes immune signaling and proliferates inside the body.
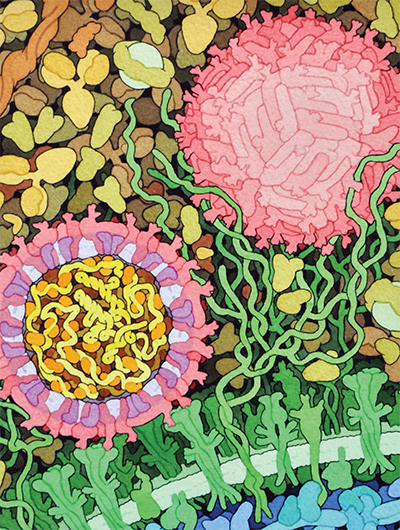 This space-fill drawing shows the outside of one Zika virus particle (red and pink) and a cross-section through another as it interacts with a cell. Courtesy of David Goodsell/Wikimedia Commons
This space-fill drawing shows the outside of one Zika virus particle (red and pink) and a cross-section through another as it interacts with a cell. Courtesy of David Goodsell/Wikimedia Commons
Like most viruses, Zika accomplishes a lot with a few tools. It has just one protein coding gene, which produces a single polypeptide that’s cleaved into 10 smaller proteins — a number dwarfed by the estimated 20,000 protein-coding genes in a human cell. Nevertheless, Zika can take over the vastly more complex human cell, repurposing it into a virus factory. Brian Raught, a researcher at the University of Toronto, said he found that process fascinating.
“With just these 10 proteins, this crazy virus turns your cells into zombies that do its bidding,” Raught said. “I always found that mind-blowing.”
Researchers in Raught’s lab, led by postdoctoral fellow Etienne Coyaud, wanted to find out how the handful of Zika proteins were able to hijack the host cell. They knew that the feat must depend on physical interactions between viral proteins and proteins native to the cell — but which ones?
Because of the increasing evidence linking Zika infections in expectant mothers to microcephaly in their children, Raught said, “We thought it better to leave the actual virus work to the experts.”
Instead of using infectious material, the team made 10 strains of human cells, each expressing one of Zika’s 10 proteins. By adding a small epitope tag to each viral protein, they were able to retrieve the viral proteins using an antibody that binds to this tag. The host proteins that stuck tightly to each viral protein came along for the ride; the researchers used mass spectrometry to identify those human proteins.
But there was a drawback to using this approach. What if proteins from the human cell were interacting with Zika proteins but separating from them before being extracted? To identify proteins that are close to but not inseparably intertwined with each viral protein, the team used a second technique called proximity labeling. In essence, they rigged each viral protein with an enzyme that would attach a sticky biotin tag onto anything that came close enough.
Proximity labeling is especially useful for detecting interactions with proteins embedded in cell membranes; those molecules are notoriously difficult to isolate.
“This is a really big asset of a proximity-labeling approach compared to traditional approaches,” Coyaud said.
Because Zika, like many viruses, enters the cell in a membrane-bound envelope and reorganizes many of its host’s membrane-bound organelles in the course of infection, its interactions with membrane proteins might be key to understanding the viral life cycle.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

The molecular orchestra of memory
Calcium, calmodulin and calcium/calmodulin-dependent kinase II form a molecular axis that turns fleeting neural activity into lasting memories. New research shows how memories are stabilized, and possibly even protected or repaired.

