
A macrocyclic lipid and the enzyme that makes it
There’s a lot to adapt to when home is a hydrothermal vent deep in the ocean. For starters, the water can be hot enough to melt a lipid bilayer.
One of many adaptations that extremophilic microorganisms called archaea make to survive their superheated, high-pressure and frequently acidic environments is remodeling their cellular membranes. Instead of two layers of lipids, archaea can link two glycerolipids by their tails to form a large, cyclic lipid called GDGT with head groups on both the intracellular and extracellular surfaces.
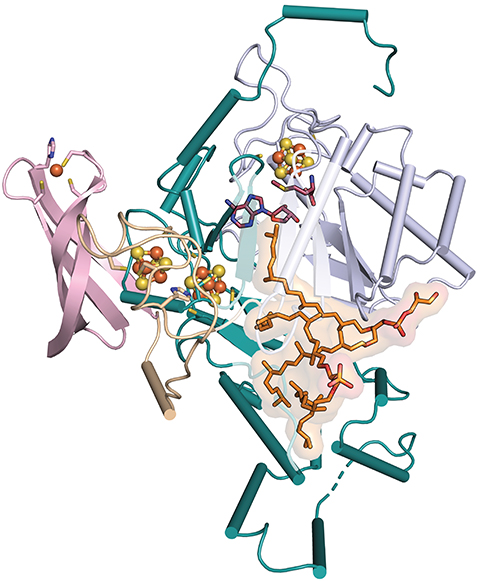
“GDGT was discovered decades ago, but no one knew what enzyme made it,” said Cody Lloyd, a graduate student at Pennsylvania State University.
Biosynthetic pathways that researchers had already worked out accounted for the lipid’s precursors but left out a single puzzling step. Somehow, the archaea must catalyze an end-to-end joining of two unreactive carbon chains. The reaction seemed to require radical chemistry, which frequently uses oxygen — but the organism in question is an obligate anaerobe.
Lloyd came across the enzyme by accident. With Amie Boal and colleagues in Squire Booker’s and William Metcalf’s labs, he reported in Nature this year its identity, its reaction mechanism and that it links the two carbons using a whole new type of radical chemistry.
While studying an enzyme believed to be a methyltransferase, Lloyd struggled to observe its reported activity. “When we solved the structure, we realized that the active site was not consistent with the proposed reaction at all,” he said.
Instead, they observed two lipids bound to the protein. Reading up on archaeal lipids, they learned about the mysterious cyclic lipid.
The researchers crystallized the protein and got it working in a test tube using a synthesized substrate. Using mass spectrometry, they captured a reaction intermediate that illuminated the enzyme’s mechanism. It abstracts a hydrogen atom from the last carbon of two saturated lipid molecules and uses an iron–sulfur cluster to stabilize one radical intermediate until the second is ready to react.
No one had ever observed an enzyme using an iron–sulfur cluster to tame a high-energy radical; the cofactor is usually involved in redox reactions. The reaction itself is also exciting for biochemists and synthetic chemists because sp3-hybridized carbons are notoriously nonreactive. Catalyzing a new bond between them is difficult in the lab and never had been observed before in nature.
“This project was thrilling,” said Squire Booker, Lloyd’s research adviser — and it stayed thrilling even when a Stanford group identified a GDGT-synthesizing ortholog a few months before the paper came out. “However, Cody’s work, besides providing the structure of the enzyme, settled a major conundrum in the field involving the nature of the substrate, and demonstrated for the first time one strategy that nature uses to link completely unactivated sp3-hybridized carbons.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Opinions
Opinions highlights or most popular articles

Learning can be fun: Gaming anatomy and physiology
Instructors explore how gamification and active learning transform student engagement and retention. They convey how emotion, interaction and design can make even rigorous subjects more effective and memorable.

Mentorship and uncertainty: Lessons from Telemachus
A biochemistry educator reflects on mentorship through the Greek story of Telemachus, showing how embracing uncertainty, failure and curiosity can transform teaching.

Embracing the twists and turns along the educator pathway
A biochemistry educator reflects on the challenges of early faculty life, describing how evidence-based teaching, cross-disciplinary collaboration and classroom challenges shaped her growth.

Redesigning with students in mind
Assistant professor reflects on how the shift to online teaching revealed gaps in points-based grading and led to a redesign centered on transparency and student growth.

Teaching beyond information transfer
Educator reflects on moving beyond lectures to create a biochemistry classroom centered on engagement, transparency and student ownership, showing how small shifts like “student hours” and active learning can transform understanding.

Mayday! Lessons from cellular dysfunction and group work dynamics
An upper-level biology course revealed that strong science doesn’t guarantee strong teamwork. One instructor shares how failed group dynamics reshaped their approach, leading to more structured, collaborative and effective student learning.

