Nautilus founder unspirals a new approach to proteomics
Parag Mallick’s latest project comes with an interesting challenge. “People keep trying to get me to say bad things about mass spectrometers,” he said in a recent interview. “You’re not going to get me to do it.”

The goal of Mallick’s company, Nautilus Biotechnology, sounds antithetical to the room-sized, half-million–dollar instruments. He intends to collect comparable data more cheaply using a benchtop instrument, proprietary reagents and an algorithm.
But Mallick, a Stanford University professor and founder and chief scientist of Nautilus, which recently went public, is the consummate mass spectrometry insider. Though his company’s mission is to make proteomics more accessible — their slogan is “Anybody who wants a proteome gets a proteome” — he argues that the expanding field can accommodate both new approaches and established mass spec techniques.
Time will tell whether his generalist attitude can be balanced with a tech company’s disruptive approach.
The technology
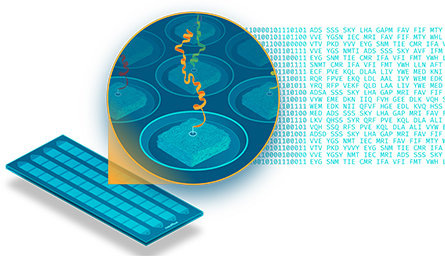
Nautilus researchers measure a proteome using a technique Mallick invented in 2016. It depends on repeated single-molecule imaging of a series of antibodies binding proteins fixed to a chip — Mallick compares it to a chessboard with 10 billion tiny squares.
Chip-based approaches to proteomics are not new; protein microarrays, in which proteins bind to antibodies adsorbed to known locations on a chip, have been in use for more than 20 years. The Nautilus approach is special because the protein sample, not the antibody probe, is fixed in place, with single proteins from the sample spaced apart by ligation to much larger polymer particles. More importantly, whereas a microarray is probed once, the proteomes Nautilus measures can be probed again and again using a line of antibodies developed in-house.
“People often do complain about their affinity reagents cross-reacting or not being specific enough,” Mallick said. “That was really the critical realization for us: to say, let’s take that concept to extremes as well, and build a class of reagents that are intentionally highly cross-reactive.”
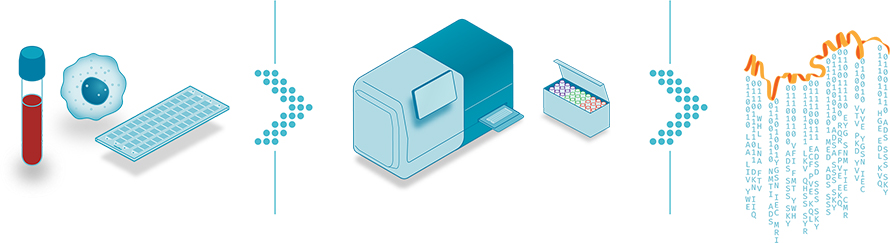
The team developed hundreds of antibodies that bind to very short epitopes, as little as a few amino acids long, that are found in many proteins. They probe with one, wash it away, and probe with the next, collecting a series of images that shows whether the molecule at each location on the chip binds with antibody 1, antibody 17 or antibody 84.
“Each one of these touches, from each one of these probes, is just a tiny little nugget of information,” Mallick said.
Those nuggets feed a machine-learning algorithm that analyzes terabytes of data per run, about 10 times as much information as is in a whole-genome sequencing run, and matches the characteristics of each spot to proteins in the proteome. The approach is particularly well suited to identifying proteoforms because of its single-molecule resolution. According to Mallick, although each binding event gives just a small amount of information about a protein, as few as 200 or 300 cycles might be enough to identify 95% of the human proteome, including specific proteoforms.
The company
Though Mallick has invented a potentially revolutionary technology and founded a company that’s already attracting big-name venture capitalists, he comes off in interviews as careful, knowledgeable and a little self-effacing. He’s an amateur magician and juggler but hid the hobby for years, concerned that his colleagues might not take him seriously as a researcher if they knew he was performing sleight of hand in his spare time.
During a recent webinar sponsored by the Human Proteome Organization, Mallick discussed Nautilus with Mark Baker, a former HUPO president and past chair of its human proteome project.
That the technology behind Nautilus would nucleate a company was not a foregone conclusion, Mallick said in the webinar. When he first had what he called “a crazy brainstorm on a weekend,” he explored several options for making it a reality, including licensing the technology to a larger company or developing it within his university lab.
Before Nautilus, he was an academic, and he’s still a professor — albeit on partial leave — at Stanford. He trained in the lab of mass spectrometry and systems biology pioneer Ruedi Aebersold, one of the most prominent researchers in the field. David Tabb, who was a graduate student at the same time in a friendly competitor’s laboratory in Seattle, said that even then, Mallick was more focused on unsolved problems than on incremental process improvements.
Later, as professors, Tabb and Mallick, along with their students Matt Chambers and Darren Kessner, co-developed a software platform called ProteoWizard that could be used to analyze mass spec data no matter what type of instrument it came from. The two labs made the software openly available, and it has been cited almost 1,400 times.
Despite that open-science background, Tabb said, it’s no great surprise that Mallick founded a company. “He’s always been smart,” Tabb said, adding that “Stanford, in general, has done a lot to incubate biotech businesses from its laboratories. … I think he had a good idea and saw a way to pursue it.”
When he was developing the idea for the new technology, Mallick said, he concluded that solving problems in biochemistry, genomics, materials science, microscopy, machine learning and other fields would move fastest in a startup. “It can be a challenge to marshal that scale in academia,” he said. “Even in the largest, most well-funded labs, you usually have a diversity of projects. We literally have every single person contributing to one goal.”
Mallick reached out for advice to contacts in the business community, including Sujal Patel, an entrepreneur and investor. Patel became a cofounder and is now the company’s chief executive officer.
“One of the lessons there is sometimes surprising things come from your network,” Mallick told Mark Baker during the HUPO webinar.
Will everyone get a proteome?
“Mass spectrometry is great for proteomics,” Tabb said. “Is it always going to be the go-to technology for identifying proteins and protein differences? I think 100 years from now, we’ll have very different answers to that question.”
According to Baker, Nautilus is making a fair bid to become one of those future answers. In an email, he wrote, “Assuming Nautilus’ platform can successfully quantify >95% of the human proteome (their vision) with a simple, fast and cheap sample prep and analysis, the concept could become as ubiquitous as sequencing a person’s genome has become today.”
Although he hopes the Nautilus platform will make proteomic experiments much more widely accessible, Mallick said that when it first rolls out, it will cost about the same per run as mass spectrometry.
Tabb, now a professor at Stellenbosch University in South Africa, said that while the Nautilus technology eventually may bring down the cost of acquiring a proteome, it is likely to remain out of reach for many researchers in the developing world. Instead, he said labs in poorer countries likely will have to find wealthier collaborators with access to instruments. That often can mean forfeiting senior authorship in order to get the experiments done. “That’s the way things have unfolded with proteomics,” he said. “That’s the way things have unfolded with sequencing.”
Whatever the cost of using a future system, it will be some time before Nautilus offers a product that any lab could buy. The company now is working exclusively with partners in the pharmaceutical industry, including Genentech, on targeted assays that do not use its custom antibodies.
“It’s very early days for Nautilus — they are only a recent spinout,” Baker wrote, while also noting, “they are moving fast, they have the backing of major tried-and-true successful investors (and) they have launched on NASDAQ.”
In the wake of that launch, the founders of Nautilus plan to double its research team and begin publishing on its approach to protein identification.

A previous version of this article misstated Parag Mallick's title at Nautilus. He is the founder and chief scientist. There was also an error in the name of Nautilus's partner company: The correct name is Genentech.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Industry
Industry highlights or most popular articles

How Alixorexton could transform narcolepsy treatment
A new investigational drug, alixorexton, targets the brain’s orexin system to restore wakefulness in people with narcolepsy type 1. Alkermes chemist Brian Raymer shares how molecular modeling turned a lab idea into a promising phase 3 therapy.

Inside industry postdocs
As more Ph.D. scientists look beyond academia, industry postdocs offer a new kind of training, where mentorship meets mission-driven research. Fellows at Pfizer and Genentech share how these programs prepare them to translate discovery into impact.

Black excellence in biotech: Shaping the future of an industry
This Black History Month, we highlight the impact of DEI initiatives, trailblazing scientists and industry leaders working to create a more inclusive and scientific community. Discover how you can be part of the movement.

Attend ASBMB’s career and education fair
Attending the ASBMB career and education fair is a great way to explore new opportunities, make valuable connections and gain insights into potential career paths.

Benefits of attending a large scientific conference
Researchers have a lot of choices when it comes to conferences and symposia. A large conference like the ASBMB Annual Meeting offers myriad opportunities, such as poster sessions, top research talks, social events, workshops, vendor booths and more.

Biotech startup worms its way into therapeutics
Andrea Choe's company, Holoclara, has created an anti-inflammatory drug based on a molecule from worms.

