Rethinking clinical trials
for next-generation cancer drugs
Cancer treatments have always been linked to a specific part of the body — these drugs for breast cancer, and those for lung cancer. Or at least they used to be.
The situation changed in May 2017, when the US Food and Drug Administration issued its first approval for a cancer drug that’s “site-agnostic.” The drug, Keytruda, had previously been approved for advanced melanoma and a few other cancers. But the new approval was remarkable because it meant that anyone with a specific genetic biomarker — no matter the initial cancer site — may use the drug.
Making treatment decisions based on a tumor’s genetic variations signifies a landmark shift in thinking about cancer. That shift has forced researchers to rethink their approach to studying new treatments. Keytruda’s approval was one of the first to follow from an emerging kind of clinical trial, called a basket trial, that evaluates a drug with a specific molecular target across many different types of cancer. These trials group patients in “baskets” according to where their tumors are located, so a single trial might include one basket for lung cancer, another for thyroid cancer and a third for breast cancer.
Basket trials are one of the first designs customized to test the idea of using genetic profiles to match patients to the drugs most likely to help them. Along the way, the trials have begun to answer questions about how rare genetic mutations contribute to cancer, says medical oncologist Gary Doherty at Cambridge University Hospitals in the United Kingdom.
Although the vision of targeted cancer therapy has remained largely out of reach, a growing number of researchers see basket trials as a way to improve the odds of success. Some early basket trials have led to some remarkable treatments, such as Keytruda, for some patients. Others have failed to find new treatments, but Doherty and others say even those trials have bolstered our knowledge about the stunning complexity of cancer, reemphasizing the diversity of its molecular underpinnings and its often dogged resistance to treatment.
New breed of trials
Mutations in more than 700 culprit genes have been implicated in the onset or growth of cancer. In any one tumor, four or five these genetic variations may turn it down a malignant path. Some mutations drive the growth of a tumor; others cripple the body’s natural defenses. There could be millions of mutation combinations — possibly even billions — behind cancerous growth. That complexity may help explain why survival rates for people with many types of advanced cancers have barely changed in the last 50 years. But at the same time, those hundreds of genes offer a tantalizing solution: more opportunities for targeted drugs to home in on vulnerabilities in a patient’s tumor.
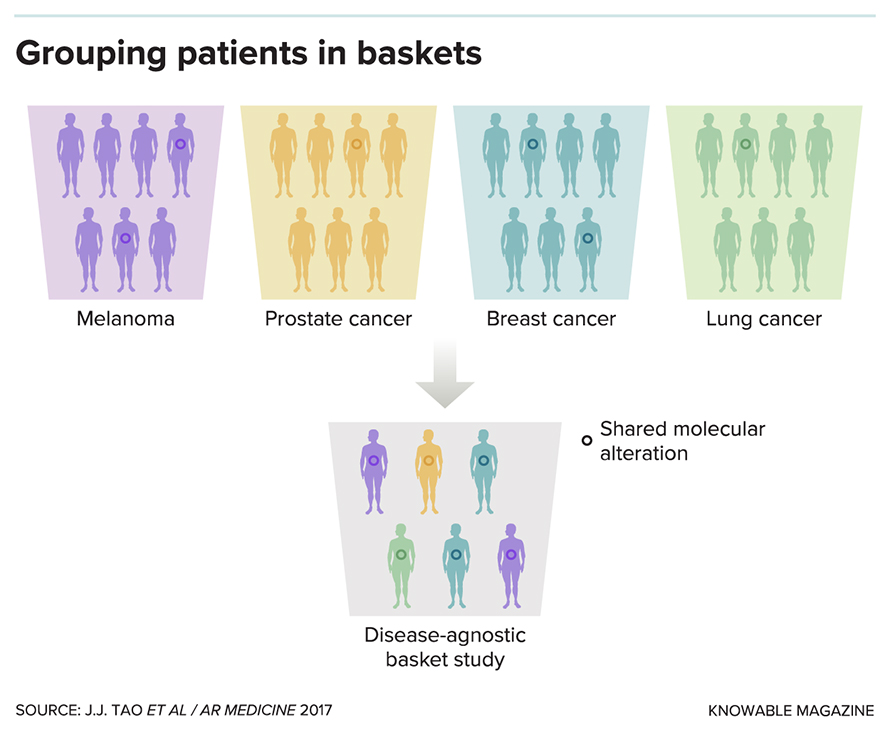
Conventional clinical trials investigate the effects of a new treatment on patients with cancer in a specific organ or tissue. Some scientists worry that such trials can mask the benefits of targeted drugs if the majority of patients don’t have the genetic mutation targeted by the drug and so don’t respond to it. One possible solution, recruiting only patients who harbor the targeted — but often rare — mutation in a particular tissue, ends up being inefficient and costly.
Using that approach, “it’s hard to enroll a clinical trial with enough patients to draw a conclusion,” says Robert Beckman, an oncologist and biostatistician at Georgetown University Medical Center. “If there are only 15 patients in the world with a disease, how can you tell with statistical significance that a therapy is working?” Beckman leads an international group of about 200 researchers focused on creating new clinical trial designs that improve the efficiency of drug development.
A basket trial gives researchers the opportunity to directly study the effect of a drug by grouping patients with cancers at a variety of sites, not just one site, based on the genetics of their tumor. “When you undertake a basket trial, you’re essentially saying that the genetic information is more fundamental than the organ site,” says Beckman.
Another advantage of basket trials is the prospect of building on the success of a therapy in one cancer type to address other malignancies. The BRAF gene, for example, is mutated in about half of all melanoma cases — and in about 5 to 10 percent of colon cancers. BRAF mutations have also been found in some lung cancers, brain cancers and non-Hodgkin’s lymphoma. (A planned phase II clinical trial is now investigating a targeted drug combination in multiple cancers with a rare BRAF mutation.)
“If you have a drug for one cancer type with a certain molecular feature, can it be used to treat other cancer types?” says oncologist Keith Flaherty at the Harvard Medical School and Dana-Farber Cancer Center in Boston, who has helped design clinical trials to answer this question.
Signals and noise
Experts point to the trial that focused on Keytruda — the first site-agnostic drug approved in 2017 — as a rousing success. Keytruda dismantles the brakes of the body’s immune cells, empowering the body’s defenses to detect and attack cancer cells. In 2013, after promising results suggesting that the drug could extend the lives of people with melanoma, an-often deadly skin cancer with few treatment options, researchers were eager to test it on other cancers. Their efforts paid off: Subsequent studies suggested that patients with some lung cancers, kidney cancers and bladder cancers might live longer with the therapy.
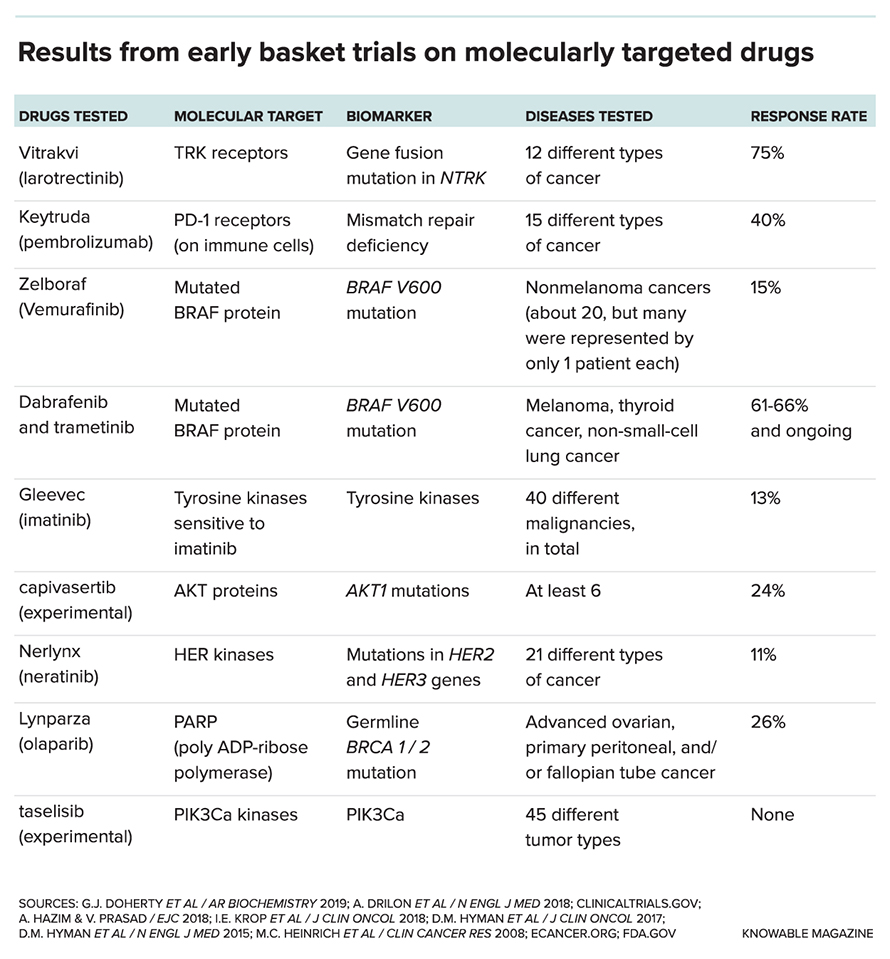
That evidence led to a basket trial that involved 149 patients with a total of 15 different cancer types. Patients qualified as long as they had mutations associated with a mismatch-repair (MMR) deficiency, a condition in which a cell’s repair shop can’t adequately fix DNA mutations connected to cancer’s growth. About 40 percent of the patients benefited from Keytruda, providing enough evidence for the FDA to grant accelerated approval — a process by which a drug can be made available to patients before all the survival data has been analyzed.
“That drug is so spectacular, and the unmet need was so great, that the FDA didn’t really require the same rigor as they demand for other drugs,” says Beckman. “It was just so evident that these drugs were very useful.”
Another success soon followed. In late 2018, the FDA approved a second site-agnostic drug, Vitrakvi, for patients who have exhausted other treatment options and whose solid tumors have what’s called an NTRK gene fusion, where a gene called NTRK breaks off one chromosome and attaches to another. The process produces what are called TRK proteins, which can promote cancer growth. That approval was based on results from 55 patients, representing 12 different types of cancer, from three small, ongoing basket trials. By eight weeks, in 12 patients, all observable signs of cancer disappeared entirely; in another 29 patients, the tumors decreased in size.
However, not all basket trials have led to effective drugs, says oncologist Aaron Mansfield at the Mayo Clinic in Rochester, Minnesota. In fact, there have been some frustrating failures.
In 2012, French researchers launched the SHIVA trial, a randomized basket trial designed to measure whether targeted cancer drugs, when used off-label, could extend progression-free survival (the amount of time after treatment until cancer gets worse). More than 700 patients were enrolled and had their tumors sequenced. About 200 patients with mutations that were targets of existing drugs were divided into two groups. About half received standard care, and the rest received a drug that wasn’t approved for their type of disease but was designed to target one of their identified genetic mutations.
The results of this large, high-profile trial were dismal. After 11 months, patients who had been prescribed targeted drugs were just as likely as the others to have cancer-related symptoms recur, and more likely to experience serious or even life-threatening side effects. That led study investigators to discourage the off-label use of targeted therapies in general.
“It didn’t move the needle” on any of the tested drugs, says Flaherty.
However, SHIVA wasn’t a total loss, says Doherty, who recently coauthored an article in the Annual Review of Biochemistry on treating cancer in the age of genomics. “The results told us that even if the same genomic biomarker in one tissue is present in another, it doesn’t mean they are both actionable,” he says. In other words, a mutation in one tumor might be driving the growth and spread of the disease, while the same mutation in another might not play as significant a role.
Mixed results have also come out of the largest basket study to date, more appropriately described as a basket-of-baskets: the National Cancer Institute’s Molecular Analysis for Therapy Choice trial, or NCI-MATCH. Flaherty is the lead investigator, and the study was designed to find promising drugs for a variety of tumor sites.
Launched in 2015, it has since enrolled more than 6,000 patients, who have each had their tumors genetically sequenced. As of early 2020, nearly a dozen separate treatment groups were still being formed, each arm of the study probing the effect of a targeted drug on a specific mutation. Where SHIVA randomly assigned targeted drugs to patients based only on a common mutation, in the MATCH trial drugs were given to patients only if previous studies in the lab and on animals suggested that they might be beneficial.
In November 2018, investigators reported that treatment with capivasertib, a drug that interferes with a cellular signaling pathway that promotes cancer growth, had so far led to tumor shrinkage in 8 of 35 patients who took it — findings promising enough to warrant future studies. But in another arm of the study, none of the 65 patients saw their tumors shrink when they took taselisib, an experimental drug that looked promising in lab and animal studies against a mutation in a gene called PIK3Ca. Other arms have had similarly weak results.
“Certainly, I’d say I’m disappointed,” says Flaherty. Knowing that taselisib doesn’t work suggests that PIK3Ca may not be the Achilles’ heel that pre-clinical trials suggested it would be, but “even a zero percent response rate tells you something,” Flaherty says.
Mixed basket
Researchers disagree about how to interpret these mixed results. One possibility is that some early basket trials suffered from flaws in their design. Many experts blame the SHIVA findings on a failure to compile enough evidence from pre-clinical animal studies to justify using the drugs tested.
More broadly, because of the way data from different treatment arms is often pooled, basket trials may not be as statistically robust as phase II conventional trials. They may also be more vulnerable to false positives, or erroneously inflating benefits. And they don’t reveal the biological significance of mutations or show why participants in one basket respond while those in another don’t.
But Doherty believes many of the obstacles can be overcome. He says that past experiences have shown that a good basket trial needs to have enough participants to make the findings reliable, a clear trial design that includes well-defined goals, and pre-clinical evidence suggesting that the drug will work for patients harboring a particular mutation.
Flaherty says basket trials, coupled with information from other genetic analyses, can fill important gaps in researchers’ understanding of which mutations do what, and when, and in which cancer types. And these pieces, he hopes, will reveal the optimal ways to get new treatments to as many patients as possible.
This article originally appeared in Knowable Magazine, an independent journalistic endeavor from Annual Reviews.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Industry
Industry highlights or most popular articles

How Alixorexton could transform narcolepsy treatment
A new investigational drug, alixorexton, targets the brain’s orexin system to restore wakefulness in people with narcolepsy type 1. Alkermes chemist Brian Raymer shares how molecular modeling turned a lab idea into a promising phase 3 therapy.

Inside industry postdocs
As more Ph.D. scientists look beyond academia, industry postdocs offer a new kind of training, where mentorship meets mission-driven research. Fellows at Pfizer and Genentech share how these programs prepare them to translate discovery into impact.

Black excellence in biotech: Shaping the future of an industry
This Black History Month, we highlight the impact of DEI initiatives, trailblazing scientists and industry leaders working to create a more inclusive and scientific community. Discover how you can be part of the movement.

Attend ASBMB’s career and education fair
Attending the ASBMB career and education fair is a great way to explore new opportunities, make valuable connections and gain insights into potential career paths.

Benefits of attending a large scientific conference
Researchers have a lot of choices when it comes to conferences and symposia. A large conference like the ASBMB Annual Meeting offers myriad opportunities, such as poster sessions, top research talks, social events, workshops, vendor booths and more.

Biotech startup worms its way into therapeutics
Andrea Choe's company, Holoclara, has created an anti-inflammatory drug based on a molecule from worms.

