Pandemic snarls research administration
Creed Stary was in a difficult position. As his research lab at Stanford gradually reopened, the physician–scientist realized he had to choose which of two grant proposals to focus on; his team wouldn’t have enough time to complete both on schedule.

Stary’s lab of two technicians and two postdocs is supported by an R01, a five-year grant of about half a million dollars from the National Institutes of Health, along with smaller individual grants to the postdocs. He needs to show the NIH continued annual progress on that major grant. In the meantime, hedging his bets, Stary also had submitted an application for a second R01 in February of this year. The pandemic upset his plans to revise that grant in response to reviewers’ concerns and resubmit it in October.
“We have to try to get the data for our existing R01, try to get the data for our new R01 revision, try to get the data for my postdoc’s grant that he’s finishing up, and also try to get the data for a new grant he’s submitting this fall,” Stary said. With limited time in the lab, his team couldn’t run all of the experiments to collect all of the data they needed on the timeline he had hoped for.
Stary wasn’t the only PI scrambling to rework his funding strategy. As the pandemic continues to beleaguer the U.S., in most of the country lab work has remained slow, stripped to the minimum hours required to keep critical research going. Now, as researchers and administrators prepare for the next set of major grant application deadlines in early October, many are especially concerned about principal investigators like Stary — relatively young, pre-tenure and vulnerable to a serious disruption if just one grant application should be denied.
The big picture
Funding to the NIH has not faltered during the pandemic. “There is a lot of money available if you’re doing research on SARS-CoV-2,” explained Ben Corb, the American Society for Biochemistry and Molecular Biology’s public affairs director, during a recent webinar hosted by the society. “If not, there is continued funding available at the same levels, and we expect an increase in fiscal year 2021.”
The Coronavirus Aid, Relief and Security or CARES Act, passed in March, included about $1.65 billion in additional funding for the NIH for COVID-19 research. As of mid-September, fiscal year 2021 budget proposals from the White House and House of Representatives call for maintained funding; and additional proposed legislation, called the Health and Economic Recovery Omnibus Emergency Solutions or HEROES Act, would add over $4 billion in supplemental research spending to the NIH’s budget if passed.
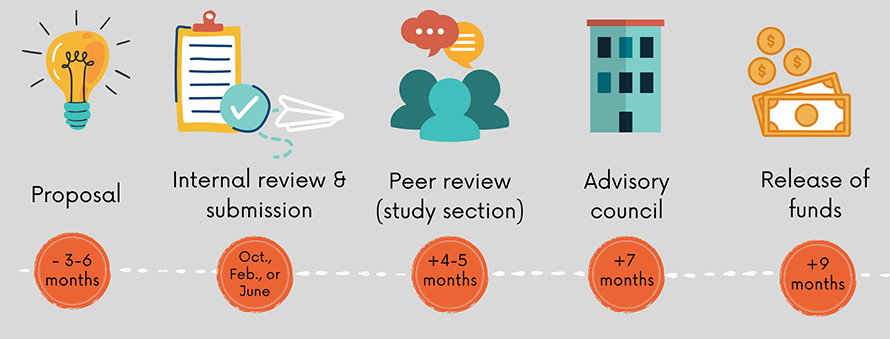
However, gaining access to that money can be a challenge. Winning research grants is a complex and time-consuming task. Up to a year can pass from when an applicant submits an investigator-initiated grant to when — if the application is successful — the funds are released. About 79% of all applications for R01 grants across the NIH are unsuccessful. Too many consecutive unsuccessful applications can force a researcher to close a lab and walk away from a research career despite decades of training.
Principal investigators spend months preparing applications and years learning grantsmanship, the unwritten rules for success in the federal funding system. They also spend significant time planning ahead to keep their labs funded continuously. Many, like Stary, are concerned about the effect of research disruptions on their funding.
Federal officials at the NIH stress that they are sensitive to these concerns and are working hard to find solutions. In a statement, the Office of External Research wrote, “We are deeply concerned and mindful about how the spread of COVID-19 has affected the biomedical research enterprise and we have implemented various administrative flexibilities.”
Some of those flexibilities, like extended deadlines to submit grants in the spring, were short-term. Others, like relaxation of strict rules for career-stage grants for postdocs and early-career independent investigators, have continued.
Research down, grant writing up
When university campuses closed across the country in March, many researchers turned to writing, data analysis and other projects to stay productive while shut out of their labs. Bill Sonntag, who directs a research center at the University of Oklahoma Health Science Center, said, “I tell the faculty in my department … ‘While we’re shut down and you have to be home, can you write a grant?’”
That flurry of writing is reflected in submission systems. Renee Gonzales, director of the Office of Sponsored Research and an assistant vice president for research at the University of Texas at Austin, said that even as other support staff faced furloughs and layoffs, her office has worked harder from home, handling expedited requests for COVID-19 research funds along with an uptick in non–pandemic-related grant applications.
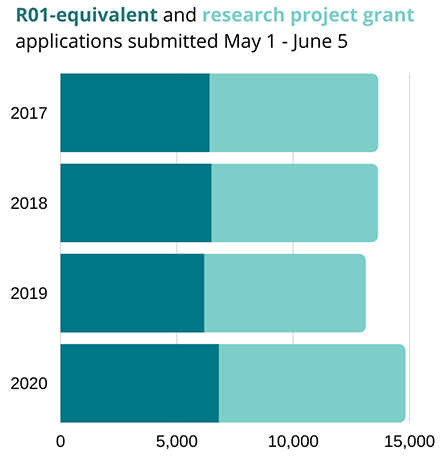
R01 and research project grant applications were both up by about 10% over
previous years in the June 2020 application cycle
Likewise, the NIH reported a 10% increase in the number of grant applications received from May to June in 2020 compared to the same funding cycle in previous years.
Phased reopenings have brought researchers back into once-shuttered labs slowly. Not all researchers were able to return to work full-time (see ‘The parent trap’). For those who could, reagents were back-ordered, animal colonies had to be reestablished, and laboratories are operating at restricted capacity to prevent crowding. The slowdown may last many more months.
The parent trap
The cost of the pandemic to research time isn’t equal across life stages. Scientists with young children lost more time than their childless peers, and mothers of young children lost more time than fathers.
Working below full capacity, researchers are trying to focus on doing the work that’s most important while also taking care to avoid starting monthslong experiments that might have to be abandoned if local public health mandates change.
“Starting out the year, you have these grand plans about planning out experiments,” Stary said. But this year, “you don’t have the luxury of all that labor that you were relying on.”
Reporting progress
At the end of each year of a funded grant, the principal investigator must submit a progress report explaining what work has been done, what money has been spent and any unexpected interruptions. University and federal program officers review that report before disbursing the next year’s funding.
Renee Gonzales’ staff typically reviews progress reports for two red flags: investigators giving less effort than expected or spending less than three-quarters of the annual budget. This year, she said, both problems are commonplace.
Oleg Barski, a program officer in biochemistry at the National Institute for General Medical Sciences, said repeatedly in an interview that the NIH has no intention of penalizing funded researchers for disruptions caused by COVID-19.
“These projects, and these PIs, were carefully selected through a very rigorous process,” Barski said. “We are here to do everything possible to ensure the success of these scientists, not to punish them for the delay.”
The progress report form leaves PIs an opportunity to explain why they made less progress or fewer purchases than planned and how they plan to address the setback. But the current environment makes it difficult to plan. Gonzales said that progress reports are full of statements such as, “‘I don’t know when I can get back up to (full) spending; I don’t know if I’m going to be able to complete the project.’ There are just so many unknowns.”
As the pandemic continues, Gonzales thinks the NIH should consider retooling the progress report. “If I feel this way in my office, I can imagine what they feel like when they get thousands and thousands of these reports. What’s the point of asking the question (about plans) when there’s no answer?”
Can I pay my technicians?
In June, the National Institutes of Health passed on guidance stating that, if grant-funded workers couldn’t work on the funded projects, universities needed to use up other sources of funding before applying grant funds to those salaries.
Early in the pandemic, Gonzales said, her office frequently fielded questions about whether researchers could keep paying their staff and their trainees (see ‘Can I pay my technicians?’). Later, other questions rose to prominence — such as what to do if money runs out before a lab can return to working at full capacity.
Even with labs closed, PIs have continued to pay personnel costs such as stipends, tuition and benefits, and most universities still charge overhead to keep lights and freezers on and pay support staff such as Gonzales’ team. Gonzales said that some researchers have expressed concern that those fixed costs, which, unlike experiments, were not interrupted, might drain grants, which could make it impossible to complete the full scope of work once their labs are back to full capacity.
If time runs out on a grant before a lab can complete the project, the investigator can apply for more time. If the money runs out, the NIH plans to make supplementary funding available — but current guidance suggests that researchers wait to apply for that boost until the pandemic’s full cost to their labs is clear.
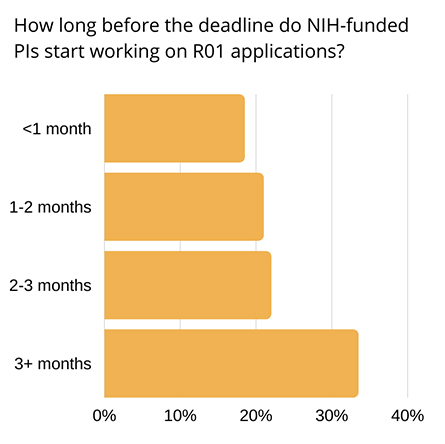
applications three months or more in advance, but many start closer
to the deadline. Respondents were polled via Twitter in September 2020.
Landing new grants
Meanwhile, the NIH continues to shepherd grants through the review process, including this spring’s bumper crop of new grants for research projects not related to COVID-19. For these new applications, some concessions were made to the pandemic. For example, researchers can submit additional preliminary data after otherwise finalizing a grant.So far, only the round of grant applications submitted, like Stary’s, before the full effects of the pandemic were felt in the U.S. have been reviewed. Stary has to convince peer reviewers, organized into a study section, that his proposed project is scientifically interesting and poised to succeed.
“They were locked at home, reading a stack of grants,” Stary mused. “I wonder if they took that into consideration, in terms of saying ‘He should do this, this and this before resubmitting’?”
According to Barski, study sections have not changed dramatically, even as they’ve gone from meeting on the Bethesda campus of the NIH to videoconferencing via Zoom. Reviewers, he said, have received instructions “to assume that issues resulting from the pandemic will be resolved prior to the award — and to show understanding that everybody has been affected.”
At the same time, reviewers are asked to be rigorous. Barski reminds researchers that they should submit the very best grant application they can muster, which may mean delaying submission by one or more cycles.
“Reviewers are still tasked to determine if the premise and feasibility of the project are solid,” Barski said. “It’s unfortunate, but if that crucial experiment, for example showing proof of principle, has not been done — and therefore the feasibility or the premise of the project is questionable — I think the PI should wait until they’re able to submit the best proposal possible.”
Reviewers generally have the prerogative to request more experiments to bolster the preliminary data that PIs present. Collecting those data can pay off; in 2019, only about 17% of grant proposals were funded after first being submitted, whereas about 33% of resubmissions made the cut after a second round of review. The study section that reviewed Stary’s grant had questions that would require more experimentation. While he initially had hoped to submit a revised application in November, he has had to accept that the lab will be unable to get it all done in time.
Meanwhile, Stary also spent time this summer reviewing grants submitted during the spring cycle. “I know how difficult it’s going to be to get the data that I would like to see,” he said a week before his deadline to submit his appraisals. He said he planned to ask himself, “What’s the absolute most critical thing they need to address here?”
According to a study section member who preferred to remain anonymous, reviewers are in a confusing position. They are not well equipped to determine how the pandemic might have affected an individual lab’s ability to collect preliminary data. Many also are concerned about whether the pandemic’s impact on different fields of research and regions can be mitigated fairly.
Ensuring continuity
Some universities have stepped in to help their professors make it through. For example, Yale and the University of Pennsylvania have introduced recovery grants up to tens of thousands of dollars to help researchers whose funding was interrupted by the pandemic or doesn’t cover costs of closing down and starting back up again.
Just how great those costs will be has yet to be seen. Kathryn Kirkland Snider, a grants administrator at the University of Colorado Boulder, said, “Given that we still don’t really have a good clear image of what the economic impact of COVID will be long term on the economy … I think it’s going to be a while before we really know the full impact (on research).”
“I’ve found that NIGMS-funded PIs are an extremely resilient and an extremely creative group,” said Barski. “Yes, I hear concerns, of course. But what I see more, is not ‘what are we supposed to do?’ but ‘what we are going to do in order to do the best possible science in the situation.’”
Although the future is uncertain, Stary, Maquat and other researchers said, in light of the economic hardship the pandemic has caused, they feel fortunate to be in a relatively stable situation for now.
“There’s a lot of heartache out there,” Stary said. “We’re among the lucky ones.”
The parent trap

challenge.
A study published in the journal Nature Human Behavior in July confirmed what many scientists report anecdotally: The cost of the pandemic to research time isn’t equal across fields or life stages. Scientists with young children lost more time than their childless peers, and mothers of young children lost more time than fathers.
School districts across the country remain closed for in-person classes. Daycare facilities are providing a fraction of their normal enrollment to limit crowding and the risk of spreading the virus. (Campus daycare options often are overbooked already, with yearslong wait lists.) And many grandparents, who can be at heightened risk from infection, are sitting out this round.
Some large universities, such as Yale, the University of California system and the University of Chicago, have invested in or beefed up backup childcare benefits for their faculty and staff and sometimes also trainees. These programs subsidize an in-home caretaker for days when a normal care plan falls through unexpectedly. However, hammering out that normal care plan is a significant challenge; young parenthood overlaps with the precarious early-career stage that lasts well into most researchers’ forties; and many are concerned that the result will squeeze parents, especially mothers, out of science altogether.
“I have one very productive postdoc who decided not to come back (to the lab) in the end,” said Lynne Maquat, a professor and director of the RNA biology center at the University of Rochester. Concerned about taking care of children and two sets of aging parents, the researcher reduced his hours to one day a week, conducted remotely, to focus on care responsibilities and begin consulting as a freelancer. Others in the lab have picked up the project he was working on.
“Can I pay my technicians?”

In June, the National Institutes of Health passed on guidance from the federal Office of Management and Budget stating that, if grant-funded workers couldn’t work on the funded projects, universities needed to use up other sources of funding before applying grant funds to continue paying salaries.
For some researchers, this was not a problem. “My people were busy,” said Lynne Maquat of the University of Rochester. Even among the scientists who were not involved in keeping animal colonies and other essential functions running, “I never had anyone furloughed in the sense that they weren’t working. We wrote commentaries, we wrote reviews, we wrote fellowships, we worked on manuscripts.”
The guidance did not apply to researchers supported by training grants, even if they could not work directly on the projects outlined in their grants. For professional staff, such as technicians and lab managers, funded from a federal source, guidelines applied across the federal government demanded that they be paid from other sources, such as laboratory startup funds, until those other sources were exhausted.
According to Stanford professor Creed Stary, the announcement was a challenge, especially since it applied retroactively. Startup money in his department generally comes from hospital income. With elective and nonemergency surgeries canceled for months, that revenue evaporated.
“The hospital was losing millions of dollars a month,” Stary said. “That profit sharing that the department can normally rely on … was essentially gone. So, suddenly everyone was very nervous about being able to support their staff.”
As far as he knows, no one working in a lab at Stanford was laid off or furloughed. But he’s concerned about year-end accounting and the effect on his lab’s startup fund, a source that many PIs rely on as a sort of savings account against a gap in funding.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Policy
Policy highlights or most popular articles

And they’re off: Budget season begins in Congress
ASBMB’s director of public affairs explains the federal budget process after the president’s proposal, highlighting proposed cuts to science agencies, next steps in Congress and how ASBMB will advocate for research funding and mobilize members.

ASBMB members advocate for funding on Capitol Hill
In more than 120 meetings with lawmakers, 50 scientists called for sustained investment in NIH, NSF and DOE research programs.

Women’s health cannot leave rare diseases behind
A physician living with lymphangioleiomyomatosis and a basic scientist explain why patient-driven, trial-ready research is essential to turning momentum into meaningful progress.

Building a stronger future for research funding
Hear from Eric Gascho of the Coalition for Health Funding about federal public health investments, the value of collaboration and how scientists can help shape the future of research funding.

Councilors advocate for science on Capitol Hill
ASBMB Councilors meet with their elected officials to advocate for basic scientific research funding and training the next generation of scientists.

Hope for a cure hangs on research
Amid drastic proposed cuts to biomedical research, rare disease families like Hailey Adkisson’s fight for survival and hope. Without funding, science can’t “catch up” to help the patients who need it most.



