JLR: Lack of sleep affects fat metabolism
We all tend to be a little short on sleep during the work week. A new study adds to the mounting evidence of how harmful insufficient sleep can be. In the Journal of Lipid Research, researchers at Pennsylvania State University report that just a few days of sleep deprivation can make people feel less full after eating and cause them to metabolize the fat in food differently.

Researchers have known for some time that sleep disruption has harmful effects on metabolism. Orfeu Buxton, a professor at Penn State and a senior author of the new study, contributed to much of the earlier research demonstrating that long-term sleep restriction puts people at a higher risk of obesity and diabetes. However, Buxton said, most of those studies focused on glucose metabolism, which is important for diabetes, while relatively few assessed digestion of lipids from food.
Kelly Ness, now a postdoctoral fellow at the University of Washington, ran the study when she was a graduate student in Buxton’s lab. After spending a week getting plenty of sleep at home, 15 healthy men in their 20s checked into the sleep lab for 10 nights, she said. After three nights spent establishing a baseline, the participants spent no more than five hours in bed for the next five nights. The researchers designed the schedule to resemble a work week.
During the study, Ness said, she and other researchers collected data, but they also spent time “interacting with the subjects, playing games with them, talking with them — helping to keep them awake and engaged and positive.”
To find out how this schedule affected metabolism, the researchers gave the participants a standardized high-fat dinner, a bowl of chili mac, once early in the study and again after four nights of sleep restriction.
“It was very palatable — none of our subjects had trouble finishing it — but very calorically dense,” Ness said.
Most participants felt less satisfied after eating the rich meal while sleep deprived than when they had eaten it well rested.
Then researchers compared blood samples from the participants. They found that sleep restriction affected the postprandial lipid response, leading to faster clearance of lipids from the blood after a meal. That could predispose people to put on weight.
“The lipids weren’t evaporating — they were being stored,” Buxton said.
The simulated work week ended with a simulated Friday and Saturday night when participants could spend 10 hours in bed catching up on missed shut-eye. After the first night, they ate one last bowl of chili mac. Although their metabolic handling of fat from food was slightly better after a night of recovery sleep, they didn’t recover to the baseline healthy level.
This study was highly controlled, which makes it an imperfect model for the real world, Ness said. It focused on healthy young people, who are usually at a lower risk of cardiovascular disease, and all the participants were men. The researchers wondered whether giving more recovery time would change the magnitude of recovery they observed.
Nonetheless, Buxton said, the study gives worthwhile insight into how we handle fat digestion.
“This study’s importance relies on its translational relevance. A high-fat meal in the evening, at dinnertime — and real food, not something infused into the vein? That’s a typical exposure. That’s very American.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition monthly and the digital edition weekly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Cows offer clues to treat human infertility
Decoding the bovine reproductive cycle may help increase the success of human IVF treatments.
Immune cells can adapt to invading pathogens
A team of bioengineers studies how T cells decide whether to fight now or prepare for the next battle.
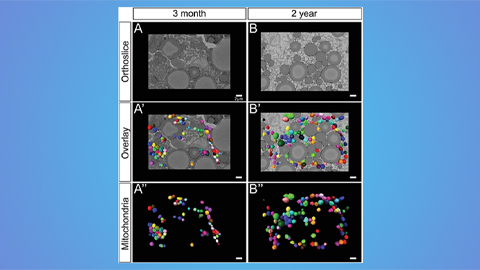
Hinton lab maps structure of mitochondria at different life stages
An international team determines the differences in the 3D morphology of mitochondria and cristae, their inner membrane folds, in brown adipose tissue.

National Academies propose initiative to sequence all RNA molecules
Unlocking the epitranscriptome could transform health, medicine, agriculture, energy and national security.
From the journals: JLR
What can you do with artificial lipoproteins? A new key to angiogenesis. Flavonoids counteract oxidative stress. Read about recent papers on these topics.
Iron could be key to treating a global parasitic disease
A study has found that leishmaniasis causes body-wide changes in iron balance, leading to red blood cell damage.

