From the journals: October 2019
We offer a selection of recent papers on a variety of topics from the Journal of Biological Chemistry, the Journal of Lipid Research and Molecular & Cellular Proteomics.
The role of ECM proteins in heart regeneration
The extracellular matrix provides a cell with mechanical and structural support and plays a role in biochemical signaling to neighboring cells. The ECM is composed of proteins including collagens, proteoglycans and glycoproteins (fibrillin and fibronectin). These ECM proteins play a role in tissue regeneration, the process by which injured tissue is restored to nearly normal function. In adult mammals, there is limited recovery of heart tissue after injury that results in a fibrotic scar, which may lead to reduced heart performance. Therefore, studying regenerative processes to design medicinal strategies is important. Nonmammalian vertebrates such as zebrafish can recover large portions of their hearts after amputation, so studying cardiomyocyte regeneration in zebra fish can help researchers design regenerative strategies for cardiovascular disease, a leading cause of death in humans.
Anna Garcia–Puig at the Center of Regenerative Medicine in Barcelona and colleagues in Spain studied the genomic and proteomic changes in ECM during heart regeneration in zebrafish. In a recent paper in the journal Molecular & Cellular Proteomics, they describe a novel method of decellularization of amputated adult zebrafish ventricles, using a detergent treatment to increase the number of ECM proteins. In comparison with older methods, this ECM protein isolation retains the integrity of ECM to provide a more robust panel of change. To identify changes in ECM proteins, the researchers decellularized zebrafish ventricles at seven, 14 and 30 days after amputation and analyzed the proteins with liquid chromatography-mass spectrometry, or LC-MS. The largest ECM protein profile changes between intact and amputated hearts occurred after seven days. Several ECM proteins — such as fibrinogen a, b and g; fibronectin 1b; and periostin b — increased after seven days, while levels of collagens and fibrillin 2b decreased.
A biomechanical property of ECM is its stiffness, which plays a role in cell proliferation, differentiation and regulation. Using atomic force microscopy, the researchers evaluated changes in zebrafish heart stiffness during regeneration. They saw a significant decrease in ventricular ECM stiffness after seven days, which returned to normal by 14 days. These data support the LC-MS findings that showed collagen was reduced after seven days; collagen concentration contributes to stiff myocardium.
— Gelareh Abulwerdi
Cholesterol transport disruption in Alzheimer’s
Scientists know that cholesterol plays a role in Alzheimer’s disease, but that role is not fully understood. In a recent study in the Journal of Lipid Research, Cinzia Marchi of the University of Parma and colleagues in Italy describe another piece of the cholesterol–Alzheimer’s puzzle.
With a growing body of work linking disruption of cholesterol homeostasis to Alzheimer’s disease, Marchi’s team asked specifically whether efflux of high-density lipoproteinlike particles is disrupted in Alzheimer’s. They examined the cerebrospinal fluid, or CSF, of Alzheimer’s patients, non-Alzheimer’s dementia patients and healthy control subjects, measuring the ability of the CSF to promote cholesterol efflux through membrane transporters. In the Alzheimer’s patients’ CSF, cholesterol efflux capacity was reduced dramatically; this was not true in the healthy controls or non-Alzheimer’s dementia patients. Future research may shed light on the intricacies of the causal role of cholesterol homeostasis in Alzheimer’s disease progression.
Refining the culprits behind Parkinson’s
Amyloid inclusions primarily composed of alpha-synuclein protein, or alpha-syn, are a hallmark of Parkinson’s disease. Different species of alpha-syn exist, but the relative contribution of each conformation to neurodegeneration has not been characterized. Jessica Froula of the University of Alabama at Birmingham and an international team produced monomers, beta-sheet oligomers and fibril fragments of alpha-syn and injected them into mouse striatum. They found that while beta-sheet oligomers exhibited some toxicity, fibril fragments produced Parkinson’s-like phenotypes, driving inclusion formation, loss of dopamine neurons and motor-behavior defects. These results, published in the Journal of Biological Chemistry, point toward alpha-syn fibrils as a potential target for Parkinson’s disease treatment.
Microbiota and obesity
Obesity and related conditions such as heart disease and type 2 diabetes are major public health problems, generating an estimated $147 billion in medical costs annually in the United States. Diet-induced obesity, or DIO, is a form of obesity with a nongenetic cause. Hao Tran at Georgia State University and colleagues in Georgia and California set out to identify biomarkers indicating proneness to DIO in gut microbiota, the diverse community of microorganisms that inhabit the gastrointestinal tract.
In their recent paper in the journal Molecular & Cellular Proteomics, the researchers looked at correlation between microbial species and protein abundance. After feeding a high-fat diet to mice, they saw a shift in the mice’s feces metaproteome. Specifically, proteins derived from species of Clostridales and Bacteroidales decreased, while proteins from Lactobacillales increased. However, they did not see this metaproteomic change when they looked at species number by fecal microbial analysis. This shows that fecal metaproteomics is a promising tool for identifying biomarkers to predict the risk of DIO after exposure to high-fat diet.
New models for the study of sarcoma treatments
The genes encoding the RNA-binding proteins FUS and EWSR1 are prone to chromosomal translocation events that produce proteins that underpin liposarcoma and Ewing’s sarcoma. Jeremy Ryan and a team of researchers at Washington University and the University of Pennsylvania report new yeast models that overexpress the aberrantly fused proteins and replicate the toxicity and aggregation patterns seen in sarcoma patients. In their study published in the Journal of Biological Chemistry, the researchers used the models to show that engineered potentiated Hsp104 proteins suppress the toxicity of the chromosomal translocations. The authors suggest that the yeast models could be useful platforms for studying both the underlying mechanisms of and treatments for sarcoma.
Whipping an acid into shape improves biofuel production
With fossil fuel shortages and carbon dioxide emissions, alternative energy sources are increasingly important. Biofuels converted from waste products like fat-rich feedstock are one alternative source, but their production is not yet efficient enough to meet the demands of many countries. Researchers in Thailand report in the Journal of Biological Chemistry that the enzyme formate dehydrogenase, or FDH, can significantly boost bacterial production of alkanes, which can be used as fuel for transportation, cooking and material synthesis.
Using synthetic biology approaches, researchers had previously designed a metabolic pathway that converts fatty acids into alkanes in bacteria. The yield of alkane from this pathway is about 35%, however, which is quite low. One reason is the limited activity of the enzyme aldehyde-deformylating oxygenase, or ADO, which carries out the final step of the process — the conversion of fatty aldehydes into alkanes with formic acid as a byproduct. If produced in excess, formic acid can decrease cellular pH, creating an acidic environment that slows ADO activity. Juthamas Jaroensuk and Pattarawan Intasian of the Vidyasirimedhi Institute of Science and Technology and colleagues aimed to increase alkane production by converting formic acid into something more useful for ADO activity.
Through bioinformatics analysis, the researchers found that the enzyme FDH from the bacteria Xanthobacter sp. 91 potentially could benefit ADO activity in two ways. Their analysis showed that FDH can remove excess formate by oxidizing it into carbon dioxide. This conversion also results in the generation of NADPH, which ADO uses to convert aldehydes into alkanes. The authors found that inserting the gene for FDH into an alkane-producing pathway expressed in Escherichia coli brought cellular pH closer to neutral and increased concentrations of NADPH. Cells expressing FDH reached conversion yields of 50%, the best yield yet for alkane production by whole-cell bioconversion.
— Jonathan Griffin
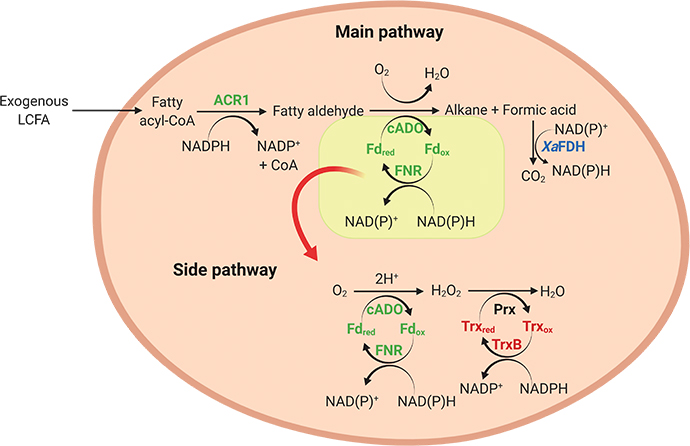 A metabolic pathway refined for enhanced alkane production: Enzymes and proteins associated with alkane production, formic acid elimination and detoxification of reactive oxygen species are shown in green, blue and red, respectively.Jaroensuk et al.
A metabolic pathway refined for enhanced alkane production: Enzymes and proteins associated with alkane production, formic acid elimination and detoxification of reactive oxygen species are shown in green, blue and red, respectively.Jaroensuk et al.
New role for the farnesoid X receptor in adipocytes
The farnesoid X receptor, or FXR, is a nuclear receptor activated by bile acids. However, research has shown that its effects go beyond the intestine. FXR is moderately expressed in adipocytes, and a new study in the Journal of Lipid Research digs into what it might be doing there.
Tim van Zutphen of the University of Groningen and colleagues in Europe and the U.S. showed that when human FXR is expressed in mouse adipocytes, a number of metabolic disease–relevant phenotypes appear, from whole-body insulin resistance to increased adipocyte size to ectopic fat deposition. Metabolic disease is increasingly common in modern society, and understanding its biology is essential to finding treatments. This study gives new insight into the complex web of interactions in metabolic diseases such as diabetes.
An unexpected metabolic target in cancer
A dependence on glycolysis over oxidative phosphorylation is thought to be a hallmark of cancer cells, but recent work has demonstrated mitochondria still are functioning in most cancers. In a report in the Journal of Biological Chemistry, Ekta Agarwal and colleagues provide new evidence for the reliance of cancer cells on mitochondrial oxidative phosphorylation. Using multiple techniques and cancer cell lines, the team showed that two oncogenic proteins, c-Myc and N-Myc, control the expression of the mitochondrial chaperone TRAP1. Moreover, they found that Myc-mediated regulation enabled oxidative bioenergetics through pathways that, when shut down within an organism, suppressed primary and metastatic tumor growth in a TRAP1-dependent fashion.
A model system to study heart development
In cell signaling, information from the exterior of the cell is transmitted to the interior. The Notch signaling pathway, one of the many signaling pathways in a cell, is conserved in many cell types and has an important role in the development of organs such as the heart.
Notch signaling plays a role in the development of heart chambers and valves, and alterations in Notch signaling can lead to cardiac disease. Early in the development of heart tissue, the heart tube is formed through outer epithelial myocardium and the inner endocardial layer. Proper signaling between endocardium and myocardium is crucial for the development of heart structures. Studying the signaling mechanism involved can help researchers understand cardiac diseases and find therapeutic interventions.
In a recent paper in the journal Molecular & Cellular Proteomics, Rebeca Torregrosa–Carrion of the Centro Nacional de Investigaciones Cardiovasculares Carlos III and colleagues in the U.S. and Spain manipulated Notch activity in mouse embryonic endocardial cells followed by mass spectrometry–based proteomics to study Notch-dependent embryonic factors secreted by heart endocardium involved in heart development and disease.
New tools to study sphingolipid metabolism
Sphingolipid metabolism is essential to many cellular processes, and this becomes obvious when defects contribute to diseases that range from developmental disorders to cancer. In a recent paper in the Journal of Lipid Research, Wataru Sakamoto of Stony Brook University and colleagues in New York and Japan describe new tools for the study of sphingolipid metabolism.
Ceramides are a family of related lipids, and the researchers’ work is framed by what they call the “many ceramides” hypothesis that the biological activity of each ceramide is determined by its localization, which is in turn determined by the localization of enzymes that metabolize ceramides. Sakamoto’s team investigated this hypothesis by developing a system to target sphingomyelinase, or SMase, which converts sphingomyelin into ceramides, and ceramidase, or CDase, which degrades ceramides into free fatty acids, to different compartments in the cell. They targeted these enzymes to the inner plasma membrane, nucleus, mitochondria, endoplasmic reticulum, Golgi and cytoplasm of HeLa cells and analyzed the lipids produced in each case.
The researchers found that SMase expression consistently increases ceramides in the cell, but the type of ceramide produced varies based on the organelle. For example, SMase in the plasma membrane acted on long-chain sphingomyelin but not on very long-chain, while SMase in the endoplasmic reticulum acted on both. They also showed that CDase expression alone does not change ceramide levels but does so in conjunction with SMase expression, which warrants further investigation.
This work sheds light on the compartment-specific effects of sphingolipid metabolism and provides new tools that can be used for future studies.
— Elizabeth Stivison
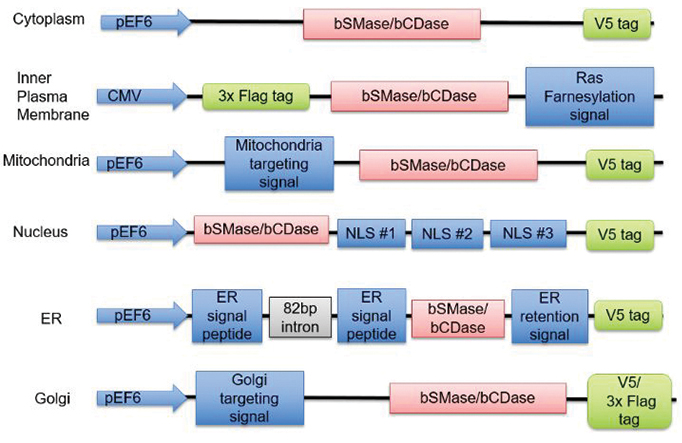 New tools created by Wataru Sakamoto and colleagues target sphingomyelinase and ceramidase to specific cellular compartments.SAKAMOTO ET AL./JLR
New tools created by Wataru Sakamoto and colleagues target sphingomyelinase and ceramidase to specific cellular compartments.SAKAMOTO ET AL./JLR
Bringing Alzheimer’s-associated proteins together
Recent reports indicate that tau protein, which forms aggregates in Alzheimer’s disease, undergoes liquid-liquid phase separation, or LLPS, but the mechanism by which this happens remains mostly unknown. In a study in the Journal of Biological Chemistry, Solomiia Boyko and colleagues at Case Western Reserve University used polyethylene glycol to induce tau LLPS and characterized the resulting droplets with fluorescence recovery after photobleaching. The authors report that tau LLPS is driven largely by electrostatic interactions and does not require phosphorylation. This insight provides a foundation to better understand how post-translational modifications could influence tau LLPS in both normal and pathological states.
Defining the birth of ribosomes
Ribosome assembly is a complex process that determines the rate of protein synthesis. Although precursor ribosomal particles, known as preribosomal complexes, have been detected in eukaryotes, their assembly remains poorly understood. Danysh Abetov and an international team of researchers present a new method to isolate and characterize preribosomes in mammalian cells. By optimizing nuclear lysis conditions, the authors preserved the complexes and characterized them with mass spectrometry and Northern blotting-based rRNA detection. They identified two new types of preribosomes and found that their formation was nutrient-dependent, elucidating a critical step in the process of ribosome assembly. The results were published in the Journal of Biological Chemistry.
Enjoy reading ASBMB Today?
Become a member to receive the print edition monthly and the digital edition weekly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
From the journals: JBC
Biased agonism of an immune receptor. A profile of missense mutations. Cartilage affects tissue aging. Read about these recent papers.
Cows offer clues to treat human infertility
Decoding the bovine reproductive cycle may help increase the success of human IVF treatments.
Immune cells can adapt to invading pathogens
A team of bioengineers studies how T cells decide whether to fight now or prepare for the next battle.
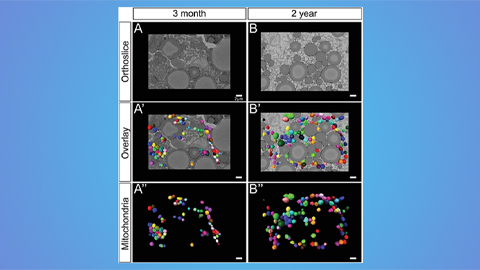
Hinton lab maps structure of mitochondria at different life stages
An international team determines the differences in the 3D morphology of mitochondria and cristae, their inner membrane folds, in brown adipose tissue.

National Academies propose initiative to sequence all RNA molecules
Unlocking the epitranscriptome could transform health, medicine, agriculture, energy and national security.
From the journals: JLR
What can you do with artificial lipoproteins? A new key to angiogenesis. Flavonoids counteract oxidative stress. Read about recent papers on these topics.



