JBC: Researchers link new protein to Parkinson’s
Researchers in Japan are reporting new insight into how the Parkinson’s disease-associated protein parkin selects its targets. The finding might improve experimental therapies for Parkinson’s that aim to boost parkin activity.
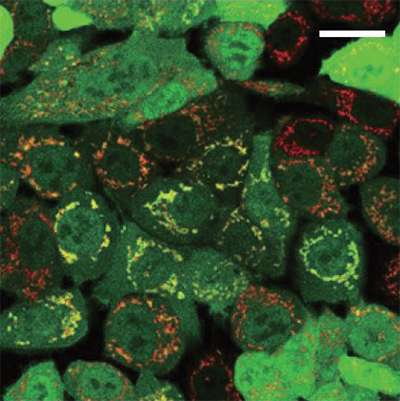
Cells depend on parkin to help get rid of damaged mitochondria. The research, published in the Journal of Biological Chemistry, suggests that parkin depends on other proteins, including one called MITOL that has not been linked previously to Parkinson’s disease, to direct it to those damaged mitochondria.
Parkin adds a degradation tag called ubiquitin to proteins on the mitochondrial surface. In some patients with familial Parkinson’s disease, parkin activity is disrupted and bad mitochondria cannot be destroyed. Harmful byproducts from those bad mitochondria can damage neurons. By understanding how parkin works and what goes wrong when it’s mutated, researchers hope also to help patients with other forms of Parkinson’s disease.
While other ubiquitin-tagging proteins, known as E3 ligases, recognize specific amino acid sequences on their substrates, parkin has many known substrates that don’t seem to share a sequence in common. While studying how parkin chooses its substrates, researchers led by Fumika Koyano in Noriyuki Matsuda’s lab at the Tokyo Metropolitan Institute of Medical Science discovered that parkin can tag any lysine-containing protein with ubiquitin — even a bacterial protein not ordinarily found in the cell — as long as it’s present at the surface of the mitochondria.
“Parkin is not regulated by its substrate specificity,” Koyano said of the finding. Instead, control of parkin activity comes from how it is recruited and activated by other proteins.
The discovery that activated parkin is not terribly selective led Koyano and her colleagues to take a closer look at parkin’s recruitment and activation. Some details of that process are well known; for example, a protein called PINK1 is known to boost parkin activity. But Koyano and colleagues discovered a new step that must happen before PINK1 can contribute to parkin activation. They found that parkin acts more rapidly when a first ubiquitin molecule is already present, acting as a seed for the addition of more ubiquitins. In most cases, the researchers found, this seed ubiquitin is added by a protein called MITOL, which had not been linked previously to Parkinson’s.
The research could contribute to the design of new drugs, some of which aim to boost parkin activity to slow the advance of Parkinson’s disease.
“If we achieve upregulation of seed ubiquitylation on mitochondria,” Koyano said, “it might accelerate parkin recruitment and parkin activation to eliminate damaged mitochondria more efficiently.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition monthly and the digital edition weekly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Cows offer clues to treat human infertility
Decoding the bovine reproductive cycle may help increase the success of human IVF treatments.
Immune cells can adapt to invading pathogens
A team of bioengineers studies how T cells decide whether to fight now or prepare for the next battle.
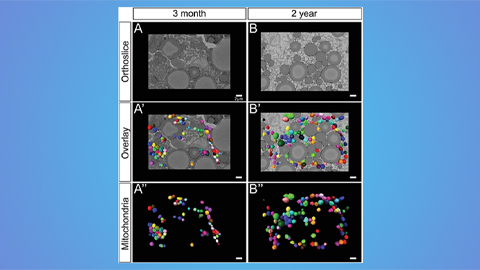
Hinton lab maps structure of mitochondria at different life stages
An international team determines the differences in the 3D morphology of mitochondria and cristae, their inner membrane folds, in brown adipose tissue.

National Academies propose initiative to sequence all RNA molecules
Unlocking the epitranscriptome could transform health, medicine, agriculture, energy and national security.
From the journals: JLR
What can you do with artificial lipoproteins? A new key to angiogenesis. Flavonoids counteract oxidative stress. Read about recent papers on these topics.
Iron could be key to treating a global parasitic disease
A study has found that leishmaniasis causes body-wide changes in iron balance, leading to red blood cell damage.

