JLR: What controls cholesterol biosynthesis?
Homeostasis is an important biochemical principle. The pace of a biosynthetic pathway often is controlled by feedback from pathway products, adjusting the system to prevent excessive accumulation of its products.
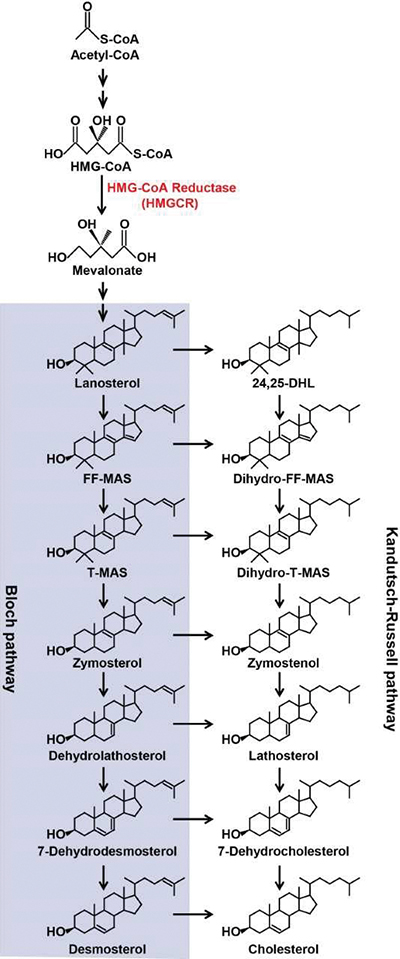 A pathway diagram shows two parallel routes from mevalonate to cholesterol.Bao-Liang Song
A pathway diagram shows two parallel routes from mevalonate to cholesterol.Bao-Liang Song
Cholesterol biosynthesis is one example. Researchers know it is regulated by metabolic intermediates but until now have disagreed about which intermediates do the work. In the Journal of Lipid Research, Liang Chen and colleagues at Wuhan University and the Chinese Academy of Sciences report that several metabolites can affect the activity of two cholesterol biosynthesis enzymes. The study gives new insight into how cholesterol biosynthesis is regulated.
Researchers knew that an intermediate product of the biosynthetic pathway could inhibit each of two control points: HMG-CoA reductase, or HMGCR, which synthesizes a key cholesterol precursor called mevalonate, and sterol responsive element-binding protein, or SREBP, a transcription factor that affects many cholesterol synthesis enzymes.
Data suggested that lanosterol, the first intermediate in the pathway that is cyclic instead of linear, was the key regulator, but the researchers knew that a slightly modified version of lanosterol might be more important. The question is complicated because the cholesterol biosynthesis pathway bifurcates after lanosterol is formed.
In lanosterol or any of its downstream products, a double bond in the molecule’s alkyl tail can be reduced, and the reduced molecules proceed through the same steps to be turned into cholesterol (see figure). So which intermediate cholesterol metabolite exerts the most control over the overall biosynthetic pathway?
That is a technically difficult problem. It is hard to induce accumulation of specific intermediates, because no effective enzyme inhibitors exist for specific steps in sterol synthesis and cells are unlikely to take up exogenously added pathway intermediates.
Chen and colleagues worked around these difficulties by generating a cell line better equipped to absorb mevalonate, a key intermediate produced by HMGCR. In these cells, intermediates can accumulate even if HMGCR activity is blocked. When these cells are provided with mevalonate, they scale up cholesterol production, triggering homeostatic degradation of HMGCR and blocks SREBP activation.
The team then systematically knocked out cholesterol biosynthesis enzymes using CRISPR, forcing traffic to back up immediately upstream of whichever conversion step had been blocked. Using lipidomic analysis of sterol extracts from each knockout cell line, they assessed the impact of loss of each enzyme and accumulation of its substrate, identifying key metabolites that impacted levels of HMGCR and SREBP.
The researchers showed that lanosterol down-regulated HMGCR but not SREBP, confirming that lanosterol and not its reduced relative is the key regulator. They also found that other sterol intermediates with reduced double bonds inhibited both HMGCR and SREBP. The authors say that molecules resembling these endogenous regulators could be a new way to control cholesterol levels.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Bacterial enzyme catalyzes body odor compound formation
Researchers identify a skin-resident Staphylococcus hominis dipeptidase involved in creating sulfur-containing secretions. Read more about this recent Journal of Biological Chemistry paper.

Neurobiology of stress and substance use
MOSAIC scholar and proud Latino, Bryan Cruz of Scripps Research Institute studies the neurochemical origins of PTSD-related alcohol use using a multidisciplinary approach.
Pesticide disrupts neuronal potentiation
New research reveals how deltamethrin may disrupt brain development by altering the protein cargo of brain-derived extracellular vesicles. Read more about this recent Molecular & Cellular Proteomics article.

A look into the rice glycoproteome
Researchers mapped posttranslational modifications in Oryza sativa, revealing hundreds of alterations tied to key plant processes. Read more about this recent Molecular & Cellular Proteomics paper.
Proteomic variation in heart tissues
By tracking protein changes in stem cell–derived heart cells, researchers from Cedars-Sinai uncovered surprising diversity — including a potential new cell type — that could reshape how we study and treat heart disease.

Parsing plant pigment pathways
Erich Grotewold of Michigan State University, an ASBMB Breakthroughs speaker, discusses his work on the genetic regulation of flavonoid biosynthesis.

