From the journals: March 2018
We offer a selection of recent papers on a variety of topics from the Journal of Biological Chemistry, the Journal of Lipid Research and Molecular & Cellular Proteomics.
Biochemical steps toward accessible vaccines

Glycoconjugate vaccines consist of a carrier protein attached to a capsular polysaccharide from the pathogen of interest. Production of glycoconjugate vaccines, for example against the bacterial meningitis agent Neisseria meningitidis, is challenging in resource-limited settings due to the biohazards and industrial-scale processes involve in purifying bacterial capsular polysaccharides. Timm Fiebig and colleagues at Hannover Medical School developed chemoenzymatic methods to produce N. meningitidis capsular polysaccharides from recombinant capsule polymerases. The streamlined protocols and optimized enzymes described in the study, published in the Journal of Biological Chemistry, were used to produce targeted carbohydrate antigens in two hours with standard laboratory equipment.
‘Minibrains’ mimic childhood disease
Sandhoff disease is a lysosomal storage disease, one of a group of rare but severe disorders caused by accumulation of macromolecules that cannot be broken down. The genetic mutations that cause Sandhoff disease affect an enzyme that breaks down the sphingolipid GM2 ganglioside. When the enzyme cannot function, GM2 ganglioside accumulates in neurons, causing seizures and cognitive and motor decline that begin at about six months of age. Most patients die before age 5. Much of what is known about the course of the disease before symptoms appear is based on animal models. In the Journal of Lipid Research, researchers at the National Institute of Diabetes and Digestive and Kidney Diseases write about using patient-derived stem cells to grow simple miniaturized brains, called cerebral organoids, which more closely model fetal brains affected by Sandhoff disease. The researchers, led by Richard Proia, compared the affected organoids with tissue from a deceased patient and with organoids with the disease mutation corrected by genome editing. As in the patient’s brain tissue taken after death, Sandhoff disease organoids showed GM2 accumulation and overgrowth; they also had delays in gene expression. The research establishes a new model for studying lysosomal storage disorders and suggests that GM2 accumulation not only causes neurodegeneration but also may disrupt neurodevelopment prior to birth.
Colon cancer and glycosylation
Cancer can develop not just because of changes in the types and amounts of proteins present in a cell but also due to changes in how those proteins are modified — for example, by attachment of sugars (glycosylation). Kirstine Lavrsen and colleagues at the University of Copenhagen discovered that GalNAc-transferase 6, known as GalNAc-T6, one of 20 polypeptides that initiate GalNAc-type O-glycosylation of proteins, was absent in healthy colon tissue but abundant in colon cancer tissue. Editing GalNAc-T6 out of a colon cancer cell line changed its growth form to one more closely resembling healthy colon tissue. Based on the glycosylation targets of GalNAc-T6 in the cancer cell lines, the authors hypothesized that expression of the enzyme disrupts epithelial development in the colon by affecting cell-cell adhesion. The study was published in the Journal of Biological Chemistry.
Babies’ skin inspires eczema lotion

Babies are soft and adorable and, as parents know, very delicate. Their skin must be moisturized frequently, because it is still slightly water-permeable. Newborns emerge from the womb covered with a waxy substance called the vernix, which protects their skin from drying. Recently, researchers have begun to realize that the vernix also helps babies adapt to life outside the womb by stimulating cells in the skin to make water-resistant lipid molecules. Scientists at Leiden University in the Netherlands, led by Joke Bouwstra, thought it might be possible to harness the vernix to treat adults with skin problems. They formulated a lotion based on lipids found in the vernix and tested it on the skin of healthy volunteers. In a recent paper in the Journal of Lipid Research, the researchers showed that disrupting the water barrier on healthy volunteers’ arms using tape caused a change in the lipids that make up the barrier. With the new, shorter-chain lipids, more water could escape through the damaged skin. Applying the lotion sped up recovery by returning the lipid profile to normal. The researchers found changes to the synthesis of lipids that were not included in the lotion, suggesting that the lotion could mimic the vernix by changing how the skin makes lipids. The researchers have not yet determined which ingredient drives the changes. The lotion, or one similar to it, might someday help treat itchy skin rashes like eczema that are driven by irritants crossing a broken skin barrier.
Comparing protein lives across species
Some proteins in the cell live longer than others. While it is established that the turnover rates of different proteins are highly variable, it is not known how conserved the turnover rates of the same proteins across species are. Researchers led by Sina Ghaemmaghami at the University of Rochester attempted to answer this question by systematically measuring the proteome turnover kinetics in primary fibroblasts from eight different rodent species, from mouse to naked mole rat. The researchers used stable isotope labeling and mass spectrometry to quantify the rate of incorporation of heavy amino acid isotopes in order to calculate protein degradation rates and half-lives. The researchers observed two striking trends. First, more closely related species have higher correlations of proteome turnover kinetics. Second, the higher the maximum lifespan of the species, the lower the global protein turnover rates. To explain the latter unexpected trend, the investigators hypothesized that long-lived species may have evolved to reduce the energetic demands of continuous protein turnover, which would lessen the generation of reactive oxygen species and the subsequent oxidative damage. This study was published in Molecular & Cellular Proteomics..
RAGE in the brain after infection
Patients recovering from sepsis often have long-term damage to the central nervous system, including cognitive impairment and neurodegeneration. Juciano Gasparotto from the Universidade Federal do Rio Grande do Sul in Brazil and colleagues at the University of Texas examined the role of receptor for advanced glycation end products, or RAGE, a signaling protein involved in both inflammation and amyloid protein function, in brain dysfunction following sepsis in rats. They found that RAGE-mediated signaling increased in brains after sepsis and appeared to increase phosphorylation of Tau protein, a hallmark of neurodegeneration. Thus, RAGE may be a key factor in the progression of long-term brain disorders after sepsis. The study was published in the Journal of Biological Chemistry.
How to IsoTaG a T cell
Post-translational modifications of proteins are important for activation of T cells during an immune response. One of these modifications, O-GlcNAc, is known to be involved in the activation of T cells; however, its function on most glycoproteins remains unknown due to difficulty in characterizing and mapping O-GlcNAc sites. In a study published in Molecular & Cellular Proteomics, investigators at Harvard and Stanford universities led by Christina Woo employed a method called IsoTaG to catalogue and quantify the O-GlcNAc sites in resting and activated human T cells. IsoTaG works by metabolically labeling O-GlcNAc residues and tagging them via click chemistry with a probe to enable enrichment and subsequent identification using mass spectrometry. The investigators identified 2,219 O-GlcNAcylated peptides from 1,045 glycoproteins, the most comprehensive characterization of O-GlcNAc modification sites so far. Using gel shift assays, they further confirmed the quantitative findings of a number of proteins that showed significant changes during T cell activation. The results provide a valuable resource for future studies aimed at a mechanistic understanding of the function of O-GlcNAc on specific proteins during T cell activation.
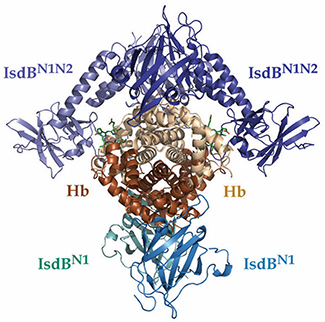 The crystal structure of the overall IsdBN1N2•Hbcomplex. IsdB molecules are colored in shades of blue, alpha Hb molecules are beige and beta Hb molecules are dark orange. Heme moieties in the alpha Hb chains are shown as green sticks.Courtesy of the University of British Columbia
The crystal structure of the overall IsdBN1N2•Hbcomplex. IsdB molecules are colored in shades of blue, alpha Hb molecules are beige and beta Hb molecules are dark orange. Heme moieties in the alpha Hb chains are shown as green sticks.Courtesy of the University of British Columbia
How staph bacteria steal our iron
Staphylococcus aureus, a widespread opportunistic pathogen, is able to “steal” heme from the hemoglobin in human blood as its preferred iron source for growth. In the Journal of Biological Chemistry, Catherine F.M. Bowden and colleagues at the University of British Columbia published a crystal structure of IsdB, a critical membrane protein in the S. aureus iron-scavenging pathway. By crystallizing an intermediate state in which heme is being transferred between hemoglobin and IsdB, the authors were able to propose a model by which IsdB unfolds hemoglobin’s heme-binding pocket to transfer heme to the bacterium.
How a kinase binds the membrane
Sphingosine-1-phosphate is a phospholipid linked to cancer and inflammatory diseases including multiple sclerosis. The phospholipid is generated when sphingosine kinase 1, called SK1 for short, translocates to the plasma membrane and phosphorylates sphingosine, but the membrane recruitment step is not well-understood. In a new paper in the Journal of Lipid Research, Michael Pulkoski-Gross and colleagues at Stony Brook University in New York identify a novel cationic patch near a known hydrophobic site on the enzyme, explaining its preference for anionic membrane lipids. Both the cationic and hydrophobic features, which form a single membrane-binding surface, are required for SK1 to bind to membranes and drive a cellular invasion phenotype that may be linked to cancer metastasis. The finding may offer a new target for drugs that disrupt the interface rather than the kinase active site.
When collagen is lost
Dystrophic epodermolysis bullosa, or DEB, is an inherited skin fragility disorder characterized by skin blistering, abnormal wound healing and excessive scarring, which often leads to aggressive skin cancer. It is caused by biallelic loss-of-function mutations in the gene COL7A1, which codes for the extracellular protein collagen VII. How the loss of collagen VII in epithelial cells contributes to DEB disease progression remains ill-understood. In a study published in Molecular & Cellular Proteomics, researchers at the University of Freiburg led by Jorn Dengjel performed a global transcriptome and proteome profiling comparing primary DEB keratinocytes to normal human keratinocytes. The researchers found that loss of collagen VII not only affected the composition of the cellular microenvironment but also led to global changes in cell homeostasis on mRNA and on protein level. They showed that TGF-beta-dependent inflammatory and proteolytic processes were perturbed in DEB cells both in vitro and in vivo. The study provides a global yet detailed picture of dysregulated molecular consequences of collagen VII deficiency.
Statins could help with wound healing
in diabetes
Statins, drugs commonly used to lower cholesterol, have additional beneficial effects, including improving wound healing. Andrew P. Sawaya and colleagues at the University of Miami published a study in the Journal of Biological Chemistry examining the mechanism by which a topically applied statin improves healing of diabetic foot ulcers, a debilitating complication of diabetes. The mevastatin treatment induced expression of a long noncoding RNA that blocked c-Myc, a transcription factor associated with nonhealing wounds. The results suggest that statins could be repurposed as part of a diabetes management regimen.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Mitochondria shape kidney cell function
Researchers at the University of Washington, Seattle present the first quantitative comparison of mitochondrial interactomes between two epithelial cell types in the kidney.

Long-chain polyunsaturated fatty acids linked to postoperative delirium risk
Researchers show that altered lipid metabolism may contribute to postoperative delirium, a condition linked to increased risk for long-term cognitive decline. The study explores potential disease mechanisms, which have yet to be understood.

Glycosylation patterns across antibody isotypes distinguish tuberculosis states
Researchers at Taipei Medical University present the first site-specific glycosylation analysis of immunoglobulins in elderly tuberculosis patients.

Blood glycome possibly predicts lifespan
Researchers at the University of Santiago de Compostela show that total serum N-glycome can predict mortality independent of traditional risk factors.

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

.jpg?lang=en-US&width=300&height=300&ext=.jpg)

