JBC: Scientists find cellular backup plan for keeping iron levels just right
Iron is essential for cell function, but excess iron can damage cells, so cells have sophisticated molecular mechanisms to sense and adjust iron levels. Disorders of cellular iron metabolism may affect more than a third of the world’s population. In addition disorders like anemia, caused by overall insufficient iron levels, iron deficiency can impair brain function in the young and reduce muscle strength in adults. Iron may be dysregulated at the cellular level in neurological disorders such as Parkinson’s disease, and disordered iron metabolism contributes to congenital conditions such as Friedrich’s ataxia.
Researchers in the nutritional sciences department at the University of Wisconsin have uncovered a connection in the network of checks and balances underlying cellular iron regulation. Their research was published in the Journal of Biological Chemistry.
When iron levels in mammalian cells are low, iron regulatory proteins, or IRPs, spring into action. IRPs prevent iron that enters cells from being improperly stored, allowing the cell to produce essential iron-containing proteins. When there is excess iron, IRPs are inactive, leading to increased iron storage, lowering potential toxicity and reserving it for when iron availability is reduced. Too much or too little IRP activity can endanger cells.
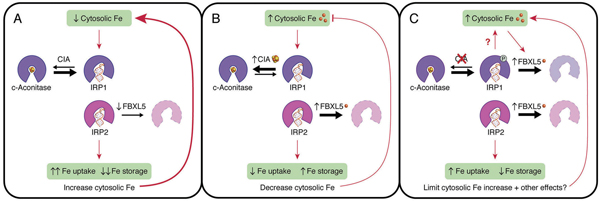 CIA, FBXL5, IRP1 and IRP2 coordinate in control of iron metabolism. Protein degradation and iron-sulfur cluster production can suppress the activity ofiron regulatory proteins, maintaining the correct iron levels in the cell. Courtesy of Caryn Outten, University of South Carolina
CIA, FBXL5, IRP1 and IRP2 coordinate in control of iron metabolism. Protein degradation and iron-sulfur cluster production can suppress the activity ofiron regulatory proteins, maintaining the correct iron levels in the cell. Courtesy of Caryn Outten, University of South Carolina
Richard Eisenstein’s research group at Wisconsin studies what controls IRP activity. Scientists have long thought the main method by which IRP-1 is inactivated involves essential compounds called iron-sulfur clusters. When there is sufficient iron in the cell, an iron-sulfur cluster is inserted into IRP-1, inactivating it. Thus, the activation or suppression of IRP-1 relates to how much iron is available to produce iron-sulfur clusters.
However, there was some evidence of another method by which IRP-1 could be stopped when it was not needed: namely, that a protein called FBXL5 could add molecular tags to IRP-1 to tell the cell to degrade the protein altogether.
“The idea that IRP1 is also regulated by protein degradation was controversial when it was first discovered by others,” Eisenstein said. “There’s been a belief that IRP-1 was really regulated by this iron-sulfur cluster mechanism, and that the protein degradation mechanism wasn’t so important.”
To test whether this was the case, Eisenstein’s team suppressed the production of iron-sulfur clusters. Even when this production was reduced, IRP-1 activity could still be suppressed. The team confirmed that this was due to the activity of FBXL5. This supported the idea that protein degradation was a backup mechanism that reduced IRP-1 action in cells with high iron.
The results have implications for understanding how iron is sensed, used and regulated in tissues. Different tissues have different levels of oxygen, but the iron-sulfur cluster production system functions best at low oxygen whereas FBXL5 functions best at high oxygen. Therefore, these two systems may trade off taking the lead in controlling IRP-1 in different parts of the body. Because iron-sulfur clusters and FBXL5 play many important roles in cell growth, this balance between these functions could help different types of cells control how they use iron.
“Diseases of iron metabolism caused by diet or by genetic perturbations are major public health issues,” Eisenstein said. “To combat such diseases and develop effective treatments for those afflicted with them, it is essential to understand iron-sensing and iron-regulatory pathways.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Building a better model for drug delivery across the blood–brain barrier
Industry and academic scientists collaborated to develop a rat with humanized iron-transport receptors, enabling research into iron homeostasis and drugs that cross the brain’s barrier.

Fat synthesis enzyme crucial for milk fat and newborn growth
Researchers found that a deficiency of the fatty acid synthesis enzyme stearoyl-CoA desaturase-1 reduced mammary gland function during lactation and caused low birth weight in newborns that were fed milk from enzyme-deficient glands.

Flipping lipids and slime molds
A dull first job nearly pushed JBC associate editor Todd Graham out of science. Then a slime mold project changed his path. Now, he studies membrane biology and reflects on discovery, persistence and mentoring through uncertainty.

How smelling death alters worm behavior
Researchers have found that the roundworm C. elegans can smell death, and it changes how the worms behave, reproduce and age.

A chance encounter with the lab
Payton Stevens never planned to become a pancreatic cancer researcher. A temporary job set him on a path from rural Kentucky to leading research on Wnt signaling and metastasis, where he now pairs discovery with mentorship and science advocacy.

Light-activated small molecule could transform eye infection treatment
Contact lenses raise the risk of infectious keratitis, a leading cause of blindness worldwide. A biotech company is commercializing a light-activated therapy using a ROS-generating molecule to rapidly kill microbes in the cornea to preserve vision.

.jpg?lang=en-US&width=300&height=300&ext=.jpg)