The cyanides of Titan
Vinyl cyanide isn’t a ’70s punk band, even though it sure sounds like it could have been. It’s a nitrogenous compound recently confirmed by NASA researchers in the journal Science Advances to exist in the atmosphere of Saturn’s moon Titan. Some scientists in the past have proposed that vinyl cyanide might have the potential to form membranelike spheres called azotosomes.
 This artist’s rendering shows the Huygens probe come to rest on one of Titan’s methane lakes. Courtesy of NASA/Gregor Kervina
This artist’s rendering shows the Huygens probe come to rest on one of Titan’s methane lakes. Courtesy of NASA/Gregor Kervina
The planetary scientists and astronomers at NASA’s Goddard Space Flight Center used calibration data obtained in 2014 from the Atacama Large Millimeter Array of telescopes in northern Chile to confirm the presence of vinyl cyanide in Titan’s atmosphere. They also estimated that quantities of the compound copious enough to reach saturation and form a solid precipitate likely exist in Ligeia Mare, Titan’s second-largest sea. Located in the north polar region of Saturn’s largest moon and named after one of the mythological Greek Sirens, the sea is about one-and-a-half times the size of Lake Superior and made up almost entirely of methane.
Titan is roughly 50 percent larger than our moon, and its frigidity is otherworldly. The average surface temperature is more than 160 degrees lower than the coldest temperature ever recorded on Earth, which is minus 128 Fahrenheit.
At such icy temperatures, cell membranes made of phospholipid bilayers like those surrounding cells found on Earth would be too rigid to function properly. A more significant barrier to life on Titan is that all known cellular processes, such as the storing of genetic information as DNA, must occur in water, meaning any biochemistry on Titan would operate in a manner entirely foreign to our own.
While the presence of vinyl cyanide in Titan’s atmosphere was predicted in 2007 in a paper in the journal Icarus based on spectral data collected by the mass spectrometer on the Cassini spacecraft, the instrument wasn’t sensitive enough to distinguish definitively between vinyl cyanide and similar compounds.
“With Cassini, they found evidence for a protonated form of the molecule in the mass spectrometer,” said Maureen Palmer at NASA Goddard, the first author on the new paper. However, she said, “you could have multiple different molecules with the same mass, so it can be hard to distinguish that way.”
Titan is the only moon in our solar system known to have a dense atmosphere and is the only celestial body other than Earth to have a dense atmosphere rich in nitrogen, which coats the planet in an orange-brown haze and falls to the surface with methane rain. In 2015, a group of researchers at Cornell University used supercomputer-generated simulations to propose that vinyl cyanide was capable of coalescing to form spherical membranes held together by the polarity of the nitrogen-containing groups, which are known as azoto groups. Thus, the structures were dubbed azotosomes.
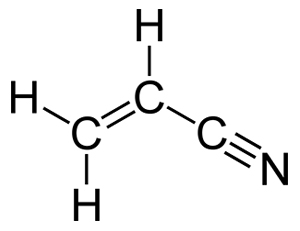 The structural formula for vinyl cyanide
The structural formula for vinyl cyanide
About cyanide
A cyanide group consists of a carbon atom covalently triple-bonded to a nitrogen atom.
When cyanide groups are attached to an organic carbon structure, they can become extremely useful for industrial applications such as the manufacturing of glues, rubbers and plastics. This is true of the double-bonded carbon atoms that make up the vinyl group in vinyl cyanide.
The infamous poisons sodium cyanide and potassium cyanide work by reacting with stomach acids to form hydrogen cyanide, which shuts down cellular respiration and ultimately cuts off oxygen to the brain.
“The key to vinyl cyanide is that it was able to form a stable, spherical structure in the liquid methane,” said Jonathan Lunine, a planetary scientist who co-authored the 2015 paper, also in Science Advances. “But, at the same time, it was flexible. Molecules that didn’t work were either ones that produced unstable spheres that would fall apart or where the spheres were completely rigid, which is not the way cellular membranes work.” If a cell membrane is too rigid, molecules are unable to diffuse in and out, making cellular processes essentially impossible.
“I was very pleasantly surprised that (the Goddard group) detected (vinyl cyanide),” said Lunine. “When we made our list of molecules that we examined in our calculations, our criterion was that they either had been detected in Titan’s atmosphere or that there was a tentative suggestion or tentative detection that they were there. That was true of vinyl cyanide. The Cassini ion-mass spectrometer had some indication in its spectra that it might be there, but it wasn’t definitive, so this was great. It’s very nice to see it’s really there.”
The presence of vinyl cyanide, though, doesn’t mean that the hypothetical azotosomes are coalescing and propagating life through strange exobiology in the seas of Titan.
“I kind of doubt that Earthlike genes and catalysts would be available,” said David Deamer at the University of California Santa Cruz’s Department of Biomolecular Engineering. “This is very conjectural, in my opinion. I like conjectures, by the way ... every conjecture can be turned into a hypothesis if you can find a way to test it.” Deamer’s research involves the origin and evolution of membrane structures.
“We’re just really depending on acts of imagination, saying, ‘Well, maybe something’s there. Let’s go look for it,’” he said. “And that’s the way science works: It’s the exploration. And these are papers exploring ideas and offering conjectural hypotheses.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Bacterial enzyme catalyzes body odor compound formation
Researchers identify a skin-resident Staphylococcus hominis dipeptidase involved in creating sulfur-containing secretions. Read more about this recent Journal of Biological Chemistry paper.

Neurobiology of stress and substance use
MOSAIC scholar and proud Latino, Bryan Cruz of Scripps Research Institute studies the neurochemical origins of PTSD-related alcohol use using a multidisciplinary approach.
Pesticide disrupts neuronal potentiation
New research reveals how deltamethrin may disrupt brain development by altering the protein cargo of brain-derived extracellular vesicles. Read more about this recent Molecular & Cellular Proteomics article.

A look into the rice glycoproteome
Researchers mapped posttranslational modifications in Oryza sativa, revealing hundreds of alterations tied to key plant processes. Read more about this recent Molecular & Cellular Proteomics paper.
Proteomic variation in heart tissues
By tracking protein changes in stem cell–derived heart cells, researchers from Cedars-Sinai uncovered surprising diversity — including a potential new cell type — that could reshape how we study and treat heart disease.

Parsing plant pigment pathways
Erich Grotewold of Michigan State University, an ASBMB Breakthroughs speaker, discusses his work on the genetic regulation of flavonoid biosynthesis.

