For better and for worse
Prion diseases — mad cow disease and Creutzfeldt–Jakob disease, for example — are downright terrifying.
You could be infected now and not even know it. The incubation period can last for decades.
You might have gotten it from an infected hamburger while in Europe years ago. Or maybe you acquired a random mutation last night while taking out the trash.
Once the infection gets going, maybe you’ll think you’re becoming a klutz. Later, you will notice a more serious tremor. Then, once amyloid plaques have turned your brain into a spongy mess, you will shake uncontrollably. A frozen, macabre smile will take over your face.
There is no cure.
That’s why it’s so easy to think of prions as evil proteins — and only that. But they’re not all bad!
As a paper that appeared in the journal Cell on Oct. 6 underscores, some prions are, in fact, advantageous.
That paper, written by a research team at Stanford University, reported the discovery of 46 new prions in yeast. The team overexpressed every gene in the Saccharomyces cerevisiae genome, stopped to allow for reproduction over many generations, and then looked to see if the progeny showed signs of that initial overexpression. They did.
The work was led by Daniel Jarosz at Stanford University. Jarosz earned his Ph.D. in Graham Walker’s lab at the Massachusetts Institute of Technology and completed his postdoctoral studies in the late Susan Lindquist’s lab, also at MIT and the Whitehead Institute. He started his own lab at Stanford in 2013.
Jarosz spoke with ASBMB Today’s executive editor, Angela Hopp. The interview has been edited for length, clarity and style.
 Daniel JaroszPHOTO PROVIDED BY DANIEL JAROSZ
Daniel JaroszPHOTO PROVIDED BY DANIEL JAROSZ
What did you study in graduate school?
I started in chemistry, but I took this class from Gerry Fink on gene regulation. It really made me think: “Wow, there are some absolutely amazing things that are happening in biology that we don’t understand mechanistically.”
One of them that excited me initially was mutagenesis. It’s clear in organisms from bacteria to humans that, in response to DNA damage, the production of mutations is an active process. It’s more than just mistakes that are made by the replicative polymerase. Actually, there are genes that you can delete and organisms won’t mutate anymore in response to the DNA damage. I wanted to understand how that works. The biochemical activity of one of those genes had been established one month before.
Graham’s lab had mostly approached this problem genetically. There were 20 years’ worth of mutants with fascinating phenotypes sitting in the freezer. It became clear to me that, now that the problem was biochemically tractable, we would be able to understand the mechanism at an unprecedented level. That’s exactly what my colleagues and I proceeded to do for several years. It was a truly exhilarating time.
So then you did a postdoc.
I think it’s hilarious that I looked for labs all over but the ideal fit was directly across the street (in Lindquist’s lab). The day that I interviewed there, someone was doing an autopsy on a patient with Parkinson’s disease. He took me on a journey, slice by slice, through the brain, looking at the deposition of protein aggregates. The day ended with meeting someone who was making amyloid fibers in vitro and then doing single-molecule optical tweezers experiments to pull on them and extract spring constants. I thought: This is amazing! It was like its own small department. There even was someone who had his own lab in Arizona and came on sabbatical (to Lindquist’s lab) and just decided he would rather remain as a senior scientist in (her) lab than go back to his own position. I was hooked by the end of the day.
Working with Susan was one of the great joys of my life. She is one of the most tenacious, brilliant and unfailingly kind people you could ever, ever imagine. (Author’s note: Later, on the day of this interview, Lindquist died of cancer.)
What did you work on there?
I developed this way to look at the traits of wild yeast strains systematically and in high-throughput. I then began looking at whether protein folding — and in particular the Hsp90 chaperone — might affect their behavior.
Another central focus of the lab was prion biology, in particular these enigmatic entities like [PSI+]. These are phenotypes that were discovered decades ago that are heritable but don’t follow Mendel’s rules, so they didn’t seem like mutations because they can be passed to all meiotic progeny, rather than half of them. They could be eliminated if you transiently treated the cells with a chemical — something that would never happen for a mutation. And they could be passed from one cell to another by mixing cytoplasm without mixing DNA. Quite unusual.
A string of really exciting studies in the 1990s established that those traits arose from prions. Since then controversy had raged over whether prions in yeast were just diseases, like PrP in mammals, or if they might have other phenotypes. Might they even be occasionally adaptive?
Several studies from Sue’s lab had taken a stab at this question, showing that [PSI+] can produce some beneficial traits. But many others were detrimental. One of the key arguments in favor of the disease hypothesis had been that when people looked for prions in wild strains, they hadn’t found them. They only had seen them in laboratory strains. With my colleague Randal Halfmann (now at Stowers Institute for Medical Research), we looked at many, many different wild strains and found that some of them did harbor prions that we knew about, including [PSI+].
But we also used this phenotyping platform to look more generally. Making no assumption about the molecular origin of the prion’s phenotype, could we test whether there were traits in wild strains that had prionlike properties of inheritance? Properties that were totally different from what you would expect for a mutation?
We found that, even in a very limited experiment, this kind of inheritance was pervasive. It was fascinating and surprising.
Contemporaneously, I got excited by another prion in wild strains — [GAR+]. And it was totally crazy.
[GAR+] regulates a switch between fermentative and respiratory metabolism. It’s conserved in (S. cerevisiae) and related species but also in fungi that are separated by hundreds of millions of years of evolution. It can be induced by a cross-chemical communication event in response to specific bacteria. And this provides benefit for both the bacterium doing the inducing and the yeast perceiving the signal. Despite this fascinating biology, [GAR+] was a real enigma mechanistically — it didn’t form amyloid fibers, for example.
This is really what motivated the work that we just published — done largely in my lab at Stanford.
Tell me about that work.
A prion is a protein that exists in multiple conformations, and one of them is like a runaway train. It can self-template once it forms. We reasoned that if we just make more protein, we should be able to increase the odds that you get a conformational switch to set that runaway train in motion.
We used an army of robots to transiently overexpress every single protein in the yeast proteome. We then asked: If we stop overexpressing and look many, many generations later, is there a molecular memory of that past overexpression?
Molecular memory?
Is there a trait that’s different in those cells whose ancestors saw overexpression compared to those cells whose ancestors either didn’t see overexpression or just saw overexpression of GFP as a control?
If we see a difference, do those traits have the same unusual genetic properties that we ascribe to prions? Do they disobey Mendel’s laws? Can they be erased with transient inhibition of chaperone activity? Can they be passed from one cell to another through cytoplasmic mixing?
We identified dozens of new prions and only three old friends. And rather than being unusual, it looks like [GAR+] is actually the founding member of a very large class of protein-based, self-templating elements that can be heritable. Most of them don’t form amyloid fibers. They don’t have these long regions of asparagine- and glutamine-rich repeats. Instead, they tend to be intrinsically disordered proteins, highly enriched in transcription factors and RNA-binding proteins.
Spell out why this is important.
Obviously, prions have been exciting to people for a long time. They motivated a Nobel Prize. (Author’s note: Stanley Prusiner won the 1997 prize in medicine or physiology for his discovery of prions.) They changed how we think about inheritance.
But people have thought of them as relatively rare. And we thought we understood what protein sequences can drive prions — again, mostly these asparagine- and glutamine-rich repetitive elements.
But [GAR+] was such an odd one out. And we could see that it was conserved for at least a couple of hundred million years.
It was found totally by chance by a lab in Canada in the late ’70s, and they had no clue what they had found. They had two papers trying to follow up on the inheritance patterns, and after it didn’t make too much sense to them, they dropped the project.
It seemed to me that the most fundamental thing about prion biology is this really unusual folding landscape — where there’s a conformation that is kinetically inaccessible but thermodynamically really stable. There’s a history to that folding landscape, where once you form the self-templating conformer, new folding events are going to be down this other pathway.
We reasoned that you could increase the likelihood that the initial misfolding event would happen just by making more protein. People have seen this before. So we had some precedent that would lead us to think that it wasn’t a totally crazy idea.
But nobody had done it on the same scale as you?
No one had done it systematically. You know, we use many other models in the lab now, but yeast remains my favorite, because you can do almost anything you want with it. For example, we could overexpress every single gene in quadruplicate, transiently, and ask: What is the effect of that — the long-lasting effect of transient overproduction.
So you overexpress and overexpress…
And then you stop. And then we grow the cells for a long time. Then we asked, at least 100 generations later: Do the grandprogeny of those mothers that saw overexpression also have a difference in their behavior? Do they have some sort of a memory of their ancestors’ experience?
How did you determine that?
Since we were doing it for many proteins, we had to be a little pragmatic. So the easiest thing to measure was simply growth rate. And we can do that in relatively high throughput. So we asked: How did those grandprogeny grow in the presence of a variety of different stressors?
So what do these prions you found look like?
They definitely don’t form fibers. Most seem to form small oligomers.
So what are the big takeaways?
First, I think, for the prion field, it suggests that a nonamyloid type of prion biology is far from rare. Rather, it may be the predominant form of protein-based inheritance. We found behavior that resembles [GAR+] far more often than we found behavior that resembles [PSI+].
Before, most everybody thought prions equaled amyloid.
Even though the biochemistry involved for an amyloid prion and these (ones we found) is very different in detail, the proteins that have this behavior have similar functions. Amyloid prions turn out to be highly enriched in proteins that regulate information flow in the cell — in proteins that are transcription factors and that that regulate the biology of RNA.
And the same is true of the proteins that we uncovered in this screen. Even though they don’t form amyloids, they don’t have the same sequence bias, etc., they are also highly enriched in transcription factors and RNA-binding proteins. It raises an interesting question.
I think people who are dyed-in-the-wool believers in prions as selfish elements probably would say that there is something about those types of proteins that are safe harbors … but an equally plausible explanation is that this is a mode of regulation that’s exploited in normal biology.
Is there anything else that our readers should know?
The other thing that is interesting is that, perhaps mercifully, given their connection to neurodegenerative disease, the N- and Q-rich sequences that drive canonical prion biology are relatively rare in our proteome. I’m fairly happy about that. Even though these could be beneficial occasionally, I’d rather not chance it too often.
In contrast, these sequences of the type that we discovered are actually more common in our proteome than they are in the yeast proteome, and indeed we, in the final experiment in the paper, were able to at least provide proof of principle that human homologues of these proteins can adopt conformations that self-template.
Are there any major misconceptions about prions?
I almost hesitate whether to call these prions at all. Prusiner, in his seminal 1982 paper, wrote what he meant by “prion:” proteinaceous and infectious. And it’s very helpful for the field that he was so clear about this definition. So I feel as if we should call them prions. But, that being said, I think, in terms of their fundamental biochemistry, in terms of their often beneficial effects on cells, it’s a loaded term. These proteins have really broadened my notion of the prion concept.
Activision and aptitude
 Daniel Jarosz with his wife, Mirna, and their two children.
Daniel Jarosz with his wife, Mirna, and their two children.
Daniel Jarosz acknowledges that he had “a maybe slightly unusual upbringing.”
He grew up in Indianola, Washington, to parents who were anti-nuclear activists. He recalls going with them, when he and his sister were little, to protest. “They would sit in front of the trains that would bring nuclear warheads to the submarine fleet that was there and wear shirts that said, ‘Only love will stop the train,’” he says. But, he adds,they’d always make sure only one of them risked arrest so that the other could look after the kids.
The elder Jaroszes, a construction worker and a housekeeper, were not alone in their activism. They were part of a small community focused on nonviolent action — “a crazy and wonderful collection of ex-hippies, nuns, priests, Buddhist monks, you name it,” as Jarosz puts it.
A product of public school, Jarosz fell for science early on. In kindergarten, he recalls, he had to fill out a questionnaire.On it, he wrote that the thing he liked least about himself was that he had collected only three fossils.
His academic potential did not go unnoticed, and he was admitted at age 14 to the University of Washington, where he double majored in chemistry and biochemistry. Going to college early was an amazing experience, he says.“I wish that more people could have the opportunity to do it.”
At UW, he initially was interested in physics, but an undergraduate research experience with a biochemist, American Society for Biochemistry and Molecular Biology member Rachel Klevit, set him on a new path.
He then enrolled in a Ph.D. program at the Massachusetts Institute of Technology. By then only 18 years old, but “sheepish” about his age, he had to decline when his peers invited him out for drinks. “I think they thought I just hated them,” he says with a chuckle. Needless to say, his social calendar was not full: “It gave me more time to focus on my work!”
But he wasn’t alone. He had a girlfriend, Mirna, who had gone through the early-admissions program at UW too.She had arrived at MIT a couple of years earlier. They’re married now and have three children.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Defeating deletions and duplications
Promising therapeutics for chromosome 15 rare neurodevelopmental disorders, including Angelman syndrome, Dup15q syndrome and Prader–Willi syndrome.
Using 'nature’s mistakes' as a window into Lafora disease
After years of heartbreak, Lafora disease families are fueling glycogen storage research breakthroughs, helping develop therapies that may treat not only Lafora but other related neurological disorders.

Cracking cancer’s code through functional connections
A machine learning–derived protein cofunction network is transforming how scientists understand and uncover relationships between proteins in cancer.

Gaze into the proteomics crystal ball
The 15th International Symposium on Proteomics in the Life Sciences symposium will be held August 17–21 in Cambridge, Massachusetts.
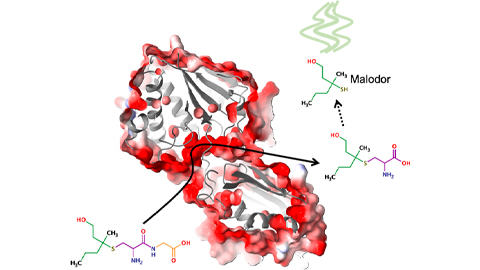
Bacterial enzyme catalyzes body odor compound formation
Researchers identify a skin-resident Staphylococcus hominis dipeptidase involved in creating sulfur-containing secretions. Read more about this recent Journal of Biological Chemistry paper.

Neurobiology of stress and substance use
MOSAIC scholar and proud Latino, Bryan Cruz of Scripps Research Institute studies the neurochemical origins of PTSD-related alcohol use using a multidisciplinary approach.

