A new test for diagnosing
Niemann–Pick disease
Niemann–Pick disease is a rare genetic disease with devastating effects. In one type of the disease, known as type C, defects in lysosomal storage within the cell lead to impaired neurological function. In infants, these symptoms can be especially difficult to recognize. They often include subtle changes in children’s development, such as failure to meet cognitive milestones or poor balance control.
Until recently, the first-line diagnostic test for NPC disease involved a skin biopsy and filipin staining, which is invasive, cumbersome and expensive. Patients with NPC often go up to five years without a diagnosis, drastically limiting the possibility of early interventions.
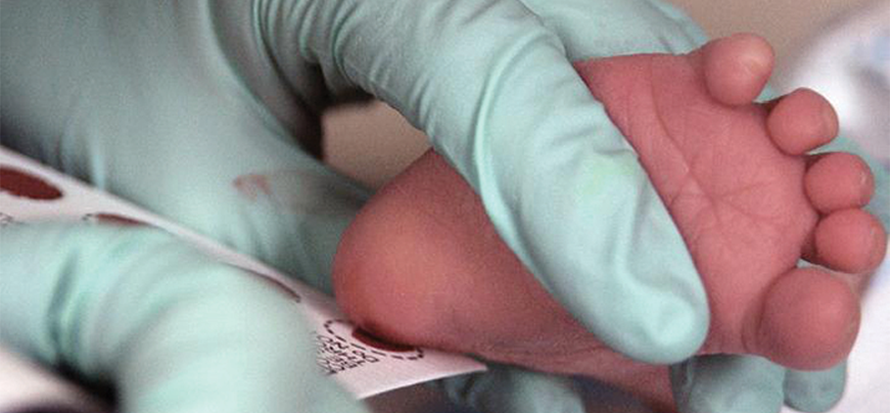 A new diagnostic test for Niemann–Pick disease can use previously collected dried blood spots from heel sticks. Courtesy of U.S. Air Force
A new diagnostic test for Niemann–Pick disease can use previously collected dried blood spots from heel sticks. Courtesy of U.S. Air Force
In a paper recently published in the journal Science Translational Medicine, Daniel Ory of Washington University School of Medicine in St. Louis and colleagues lay the groundwork for a promising new diagnostic test for NPC. Importantly, the new noninvasive assay produces results within a day instead of months.
The team used mass spectrometry to analyze dried blood spots collected at various times after birth from patients known to have NPC. They found three bile acid biomarkers that could distinguish NPC patients from people without the disease.
The scientists then determined the structures of the bile acids. Ory and colleagues identified one bile acid as a trihydroxycholanic acid and another as its glycine conjugate.
Since the second bile acid helped the team distinguish NPC patients from non-NPC patients more consistently, the researchers decided to use it to develop a new diagnostic test.
Ory says the assay already is being used at Washington University in St. Louis, Mo., as a diagnostic test. He expects other centers to follow suit.
For its use in newborn screening, Ory says researchers will need to put the assay to the test in the undiagnosed newborn population to ensure its usefulness for that age group. Ory believes the testing process will take several years.
Although the U.S. Food and Drug Administration hasn’t yet approved treatments for NPC, a promising drug called cyclodextrin is moving rapidly through clinical trials. To be effective, treatment interventions will need to take place early in the disease process, which is something the new test could help accomplish. “We’re really trying to make an impact in this NPC community by being able to develop the therapies and being able to diagnose early,” says Ory. The approach “we’ve taken over the last 10 years, I feel like, it’s getting close to bearing fruit.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles

Defeating deletions and duplications
Promising therapeutics for chromosome 15 rare neurodevelopmental disorders, including Angelman syndrome, Dup15q syndrome and Prader–Willi syndrome.
Using 'nature’s mistakes' as a window into Lafora disease
After years of heartbreak, Lafora disease families are fueling glycogen storage research breakthroughs, helping develop therapies that may treat not only Lafora but other related neurological disorders.

Cracking cancer’s code through functional connections
A machine learning–derived protein cofunction network is transforming how scientists understand and uncover relationships between proteins in cancer.

Gaze into the proteomics crystal ball
The 15th International Symposium on Proteomics in the Life Sciences symposium will be held August 17–21 in Cambridge, Massachusetts.
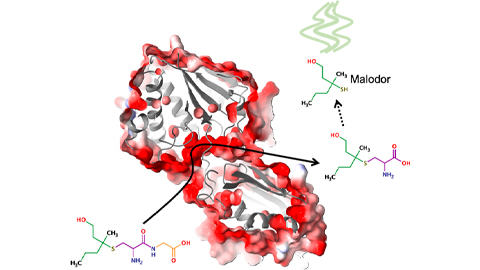
Bacterial enzyme catalyzes body odor compound formation
Researchers identify a skin-resident Staphylococcus hominis dipeptidase involved in creating sulfur-containing secretions. Read more about this recent Journal of Biological Chemistry paper.

Neurobiology of stress and substance use
MOSAIC scholar and proud Latino, Bryan Cruz of Scripps Research Institute studies the neurochemical origins of PTSD-related alcohol use using a multidisciplinary approach.

