JBC: Probing beneath the surface
In the early 1980s, when first-year graduate student Carmen-Rosa Torres brought Gerald Hart the first evidence of O-linked N-acetylglucosamine, or O-GlcNAc, he was sure the data were wrong. Only after making Torres repeat the experiments for a year and jumping into the lab himself was Hart finally convinced. Torres and Hart’s discovery of O-GlcNAc was published in 1984 in the Journal of Biological Chemistry.
O-GlcNAc is an oligosaccharide bound to a hydroxyl moiety on a serine or threonine residue of a protein. It’s formed when the enzyme O-GlcNAc transferase transfers a GlcNAc moiety from UDP-GlcNAc to a specific amino acid residue, a process known as O-GlcNAcylation.
In the 1980s, scientists were confident that all glycosylated proteins either were transported outside of the cell or were confined to intracellular compartments after processing. The 1986 JBC paper that came out of Hart’s laboratory at the Johns Hopkins University School of Medicine laid out the first proof of O-GlcNAc in the cytosol and nucleus. The discovery described in the paper, now recognized as a JBC Classic, violated the reigning dogma of the time. Even within the glycobiology community, it was hard to accept the idea that glycoproteins would be functioning inside the cell.
“Frankly, it was controversial enough that, when we presented the findings at conferences, I had people basically saying to my face that they didn’t believe the results,” says Gordon Holt, who was a graduate student in Hart’s lab and a co-author on the 1986 paper. (Torres was working on another project at that time.)
 In 2014, Gerald Hart, an associate editor for the Journal of Biological Chemistry, organized a thematic minireview series on O-GlcNAcylation.
In 2014, Gerald Hart, an associate editor for the Journal of Biological Chemistry, organized a thematic minireview series on O-GlcNAcylation.
O-GlcNAc is found on many different proteins throughout the cell. Its function varies dependent upon the protein to which it is attached. Much like phosphorylation, one known purpose of O-GlcNAc is to control signaling in response to nutrients. Holt, who is now the chief science officer at NorthShore Bio, describes O-GlcNAc as an orthogonal method of regulating the same proteins: “It’s like a rivet: If there’s a carbohydrate at a phosphorylation site, then that phosphorylation site is no longer regulated by classical phosphorylation pathways. Instead, it becomes regulated by the O-GlcNAc regulatory pathways.”
Methods for studying phosphorylation have advanced rapidly in recent years, thanks in part to the existence of site-specific antibodies, while methods for studying O-GlcNAc continue to lag behind. Researchers only recently have begun to recognize the importance of O-GlcNAc to chronic diseases. Prolonged elevation of O-GlcNAc, as in hyperglycemia, can cause transcriptional processes to go haywire.
“It’s something that we have to understand mechanistically very carefully, because it is essential for understanding lots of the intracellular signaling pathways and the way the cell can function when glucose goes up or down,” says glycobiologist Vincent Hascall at the Cleveland Clinic.
Hart says a lot of the deleterious effects of diabetes are caused by elevated O-GlcNAc, and that “O-GlcNAc has also been elevated in every cancer type that’s been examined to date.” Low levels of O-GlcNAcylation in the brain also have been linked to Alzheimer’s disease. Several drug companies are developing treatments that inhibit O-GlcNAc hydrolase, the enzyme governing O-GlcNAc removal.
In their 1986 experiment, Hart and Holt used highly purified fractions of cells to pinpoint where the bulk of O-GlcNAc modification was located. Hart remembers sitting Holt down to break the news that preparation for the experiment would take him close to a year. Holt listened and nodded and then grabbed an ice bucket and went around to different labs at Johns Hopkins, collecting all the organelle samples needed — what Holt calls his “cup of sugar in the form of organelles” — from some of the leading experts in organelle purification. The preparation was completed in just a few days. “It was pretty amazing,” says Hart. “He had a graduate student network. They know what’s going on in the place way better than the faculty do.”
Faculty members and students teased Holt for taking a shortcut. “It occurred to me, in retrospect, that the ice-bucket strategy was the path of least resistance, but it was also critically important, because it got us unquestionably pure organelles,” says Holt. “Our results needed to be taken seriously as a result.”
After obtaining the samples, they attached radioactive sugar residues to galactosyltransferase, an enzyme purified from cow’s milk. They used radioactive tags to identify which proteins the enzyme attached to in each organelle.
“The prevailing opinion was that intracellular cytosolic proteins were not glycoproteins,” says Hascall. “They showed in this paper, which opened up a whole new field, that that was not true. There is, in fact, a very specific glycosylation on a number of intracellular proteins.”
“It’s not very often in your career that you run across something completely novel and totally unexpected,” says Hart. “When you do, you should probably follow your data.”
This article originally was published in the Journal of Biological Chemistry.
Enjoy reading ASBMB Today?
Become a member to receive the print edition four times a year and the digital edition monthly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
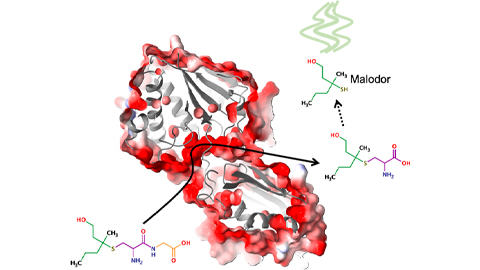
Bacterial enzyme catalyzes body odor compound formation
Researchers identify a skin-resident Staphylococcus hominis dipeptidase involved in creating sulfur-containing secretions. Read more about this recent Journal of Biological Chemistry paper.

Neurobiology of stress and substance use
MOSAIC scholar and proud Latino, Bryan Cruz of Scripps Research Institute studies the neurochemical origins of PTSD-related alcohol use using a multidisciplinary approach.
Pesticide disrupts neuronal potentiation
New research reveals how deltamethrin may disrupt brain development by altering the protein cargo of brain-derived extracellular vesicles. Read more about this recent Molecular & Cellular Proteomics article.

A look into the rice glycoproteome
Researchers mapped posttranslational modifications in Oryza sativa, revealing hundreds of alterations tied to key plant processes. Read more about this recent Molecular & Cellular Proteomics paper.
Proteomic variation in heart tissues
By tracking protein changes in stem cell–derived heart cells, researchers from Cedars-Sinai uncovered surprising diversity — including a potential new cell type — that could reshape how we study and treat heart disease.

Parsing plant pigment pathways
Erich Grotewold of Michigan State University, an ASBMB Breakthroughs speaker, discusses his work on the genetic regulation of flavonoid biosynthesis.

