From the journals: MCP
What do oncogenes regulate? Immunopeptidomics keeps teaching us. Read about articles on these topics recently published in Molecular & Cellular Proteomics.
What do oncogenes regulate?
Oncogenes are genes that have the potential to cause cancer. Depending on the oncogenes’ functional role, their mutations, overexpression or downregulation drive various forms of cancer in humans. And development of cancerous tumors results in massive multiomics changes. Researchers seek to understand these molecular changes to identify unifying hallmarks and broader drug targets more effectively.
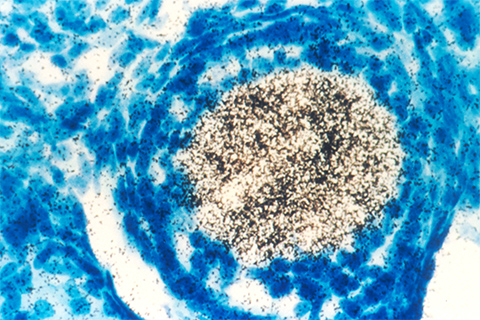
Paige E. Solomon and researchers at the University of California, San Francisco, and the University of Wisconsin–Madison, studied these changes using a proteomics approach. Looking at isogenic cell lines with or without seven driver oncogenes, known as MYC, EGFR, HER2, AKT, KRAS, BRAF and MEK, they found that oncogene expression in each directly caused up- and down-perturbations in proteomes, compared to the corresponding cell line without the oncogenes. In particular, the researchers noticed strong and universal downregulation of several antiviral pathway and type 1 interferon pathway proteins including IFN beta, all of which are essential in regulating immune responses.
Oncogene expression also dramatically affected dsRNA sensing pathway proteins. However, dsDNA sensing proteins remained relatively unchanged, leading the researchers to predict that dsRNA sensing could have functional consequences that are distinct from the dsDNA sensing signaling pathways in cells expressing oncogenes. They found that cells expressing oncogenes have impaired dsRNA-sensing antiviral responses and increased susceptibility to RNA viruses. Their findings recently have been published in the journal Molecular & Cellular Proteomics.
As cells with oncogenes show increased susceptibility to RNA viral infection, researchers potentially could target tumors using virotherapy. Moreover, in many cancers, low interferon expression indicates aggressive and drug-resistant subtypes. Knowing that oncogene expression directly suppresses interferon proteins could help researchers develop therapeutic targets for aggressive forms of cancer.
Immunopeptidomics keeps teaching us
Immunopeptidome research originated with studies of the genetics of tissue transplantation, which showed that the major histocompatibility complex, or MHC, is a critical mediator of transplant survival. The MHC on immune cells, such as T cells, contains a set of genes that code for cell surface proteins essential for the adaptive immune system.
Based on the nature of the antigens presented to the MHC molecule, these T cells can distinguish between what researchers refer to as “self” and “nonself.” Recognition is based on MHC class I proteins (which present endogenous antigens that originate within the cell) and MHC class II proteins (which present exogenous antigens, such as viruses and bacteria that are present in body fluids).
The immunopeptidome broadly refers to the peptides presented by MHC molecules on the surface of antigen-presenting cells to enable T-cell immunosurveillance. In a recent Molecular & Cellular Proteomics perspective article, Jonathan W. Yewdell of the National Institute of Allergy and Infectious Diseases provides a comprehensive review of the past, present and future of immunopeptidome research. In chronicling the history of this field, he outlines the biochemical studies that explain the MHC class I–restricted nature of T-cell recognition, which ultimately culminated in seminal studies using mass spectrometry to characterize host cell class I–bound and class II–bound peptide ligands. He describes how MS has become a critical technique for defining MHC-associated peptides and understanding how peptides are generated from the antigen-presenting cells.
Yewdell also discusses future contributions of MS to the understanding of the MHC immunopeptidome. Increased MS sensitivity could characterize the immunopeptidome at the single-cell level, and future research could obtain quantitative data from MS studies and use the MS immunopeptidome to characterize contribution of antigen-presenting peptides to cellular processes and diseases.
Enjoy reading ASBMB Today?
Become a member to receive the print edition monthly and the digital edition weekly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Iron could be key to treating a global parasitic disease
A study has found that leishmaniasis causes body-wide changes in iron balance, leading to red blood cell damage.

Environmental DNA is everywhere
The ability to extract trace bits of DNA from soil, water, and even air is revolutionizing science. Are there pitfalls?
Early COVID-19 research is riddled with poor methods and low-quality results
The pandemic worsened, but didn’t create, this problem for science.

From the journals: MCP
Three views of mass spec: analyzing secreted protein spectra, imaging mass spectrometry for clinical use and spectral libraries for MS data analysis. Read about these recent papers.

Understanding the fat science
Researchers at UCLA investigate lipid remodeling in the liver for energy generation.

No oxygen? No problem
By studying how electric fish survive in hypoxic streams for months at time, researchers may find new ways to target tumors.

