From the journals: September 2019
Picking cancerous mutations out of the crowd
Mutations that drive cancerous behavior may be candidates for targeted therapy, but differentiating them from the thousands of inconsequential mutations in cancer tissues has remained a challenge. Deepankar Chakroborty and an international team of researchers devised a new screening method to show cancer-associated mutations. They used the ability of Ba/F3 lymphoid cells to overcome their dependence on interleukin 3 with activated epidermal growth factor receptors, or EGFRs, which are perennially turned on in many cancers. By introducing random mutations into EGFRs and starving cells of IL-3, only those with EGFR-activating mutations survived.
This technique, presented in the Journal of Biological Chemistry, offers a simple means of identifying EGFR variants that potentially drive cancerous activity and merit further investigation.
Fat in muscle linked to insulin resistance
Diets high in saturated fat are associated with metabolic diseases. Insulin resistance caused by fat depletion, known as lipodystrophy, is rare but has become a focus of research, including studies of associations between accumulation of fats stored in muscle, known as intramyocellular lipids, and insulin-resistant or insulin-sensitive states. The amount of intramyocellular lipid has been associated with both states (known as the athlete’s paradox).
David B. Savage, Alison Sleigh and researchers from the U.K. and Switzerland write in the Journal of Lipid Research that they used magnetic resonance spectroscopy to compare the fatty acid composition in calf muscles of insulin-resistant lipodystorphic women with age- and gender-matched healthy controls and athletes. They hypothesized that differences in composition of the intramyocellular lipid pool might explain the athlete’s paradox. They found that the intramyocellular lipid pool was more saturated in the lipodystrophic women compared with controls and athletes. Measures of the accumulation of saturated intramyocellular lipid were more strongly associated with measures of insulin sensitivity than the total amount of intramyocellular lipid.
A strong correlation in athletes and healthy controls between maximum oxygen used during exercise and relatively unsaturated intramyocellular pools suggests exercise and diet can help decrease the amount of saturated fat within muscles associated with insulin resistance.
One step closer to fertilizer-free farming
Farmers apply fertilizer to deliver ammonia and other essential nutrients to crops, but excess ammonia can wash away into fresh water, a hazard for wildlife and humans. To reduce agriculture’s dependence on fertilizer, scientists seek to enhance the ability of plants to provide for themselves through nitrogen fixation, a process that converts nitrogen from the air into ammonia. One method is getting aerobic plants to express nitrogen-fixing enzymes, but oxygen deactivates these enzymes, known as nitrogenases. In a study in the Journal of Biological Chemistry, researchers in the U.K. biochemically and structurally characterized a protein that might protect nitrogenases from oxygen inactivation.
In nitrogen-fixing microorganisms, three types of nitrogenases have been identified; each relies on a different metal to function. The iron-only nitrogenase is the least understood of the three, but scientists view it as a promising candidate for expression in plants, as this enzyme requires fewer genes than the more common molybdenum nitrogenase. Other researchers previously had found that the purple bacterium Rhodobacter capsulatus requires expression of the protein Anf3 for its iron-only nitrogenase to work, but researchers didn’t know why. To solve this mystery, Febin Varghese and colleagues at Imperial College London examined the structure and function of Anf3 in the model nitrogen-fixing bacterium Azotobacter vinelandii.
Using X-ray crystallography to view Anf3’s structure at an atomic resolution, the team saw that it was similar to a family of oxygen-reducing enzymes. Electrochemical experiments showed that Anf3 converts oxygen to water, confirming its oxygen-reducing activity. These results indicate that Anf3 may protect iron-only nitrogenase from deactivation by consuming oxygen, which could be why the protein is needed for nitrogen fixation in R. capsulatus. The findings also might mean that Anf3 could do the same in aerobic plants, bringing nitrogen-fixing crops closer to reality.
— Jonathan Griffin
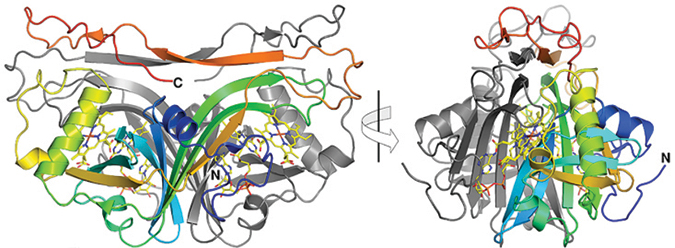 In these orthogonal views of the dimeric, two-chain structure of Anf3, the A chain is shown in multiple colors, and the B chain is shown in gray.Varghese et al.
In these orthogonal views of the dimeric, two-chain structure of Anf3, the A chain is shown in multiple colors, and the B chain is shown in gray.Varghese et al.
Potential drug target for Parkinson’s
Research suggests that chemicals in the brain similar to the neurotoxin MPTP may induce or enhance the progression of Parkinson’s disease. In 2013, a team of researchers from Vanderbilt University and the University of Pennsylvania found that the enzyme cytochrome P450 2D6, or CYP2D6, in the mitochondria metabolizes MPTP into a toxic form. The same team recently used dopamine neuron-mimicking cells and mice with the Cyp2d6 gene knocked out to show that the conversion of MPTP-mimicking chemicals produced in the brain by CYP2D6 induces neuronal degeneration. The results, published in the Journal of Biological Chemistry, suggest that CYP2D6 may be a promising target for treating Parkinson’s disease.
How smoking triggers changes in Reinke’s edema
Among the negative effects of smoking is Reinke’s edema, or RE, where swelling of the vocal cords causes breathing and voice problems. Treatment involves surgical stripping of vocal cord layers in a section called the lamina propia, which often results in permanent damage. In the search for better treatment options, vocal fold fibroblasts, or VFFs, the main cellular component of the lamina propia, have become a focus of RE research. To better understand the effect of smoking on these cells, Markus Gugatschka and his colleagues at the Medical University of Graz exposed VFFs to cigarette smoke and monitored the subsequent protein changes.
In a paper published in the journal Molecular & Cellular Proteomics, the researchers write that they found increased levels of the inflammation-promoting polysaccharide hyaluronan and antioxidative stress proteins as well as lower levels of the rigid structural protein collagen. The researchers hypothesize that the loss of collagen, increase of hyaluronan and increased VFF cell death from oxidative stress could combine to facilitate the formation of edema, thus leading to RE. Future research to evaluate this hypothesis could provide potential targets for treatments to combat RE.
The role of LOXL2 in prostate cancer
An early step in the development of prostate cancer involves alteration of tissue-building cells called fibroblasts. Normal (healthy) prostate fibroblasts, or NPFs, respond to signals in the tumor to become cancer-associated fibroblasts, or CAFs. These CAFs produce proteins that stimulate production of the extracellular matrix, or ECM, which provides structural support and aids tumor growth. Despite the known importance of CAFs in prostate cancer, CAF–ECM interactions remain poorly understood. To understand these interactions better, Elizabeth V. Nguyen, Brooke A. Pereira and colleagues in Australia sought to characterize the protein differences between prostate CAFs and NPFs.
In their recent work, published in the journal Molecular & Cellular Proteomics, the researchers isolated CAFs and NPFs from four patients with prostate cancer and compared their respective protein profiles. They found that CAFs produced more proteins responsible for cell adhesion, specifically through affecting collagen (a major structural element of the ECM). One of these proteins, LOXL2, has previously been shown to be more prevalent in prostate cancer, and the authors showed that this higher prevalence was associated with poorer prognosis. Further study on LOXL2 showed that this protein’s overproduction by CAFs not only led to a more structured and robust ECM but also aided in the growth and enhanced the health of prostate tumor cells.
This is the first time scientists have shown directly that LOXL2 inhibition can perturb prostate tumor development. Previous research has shown that LOXL2 is amenable to therapeutic targeting, and drugs targeting LOXL2 already are being evaluated for use against breast and pancreatic cancer. LOXL2 shows significant promise as a target for more oncology therapies, and these researchers’ work supports this approach.
— Kian Kamgar–Parsi
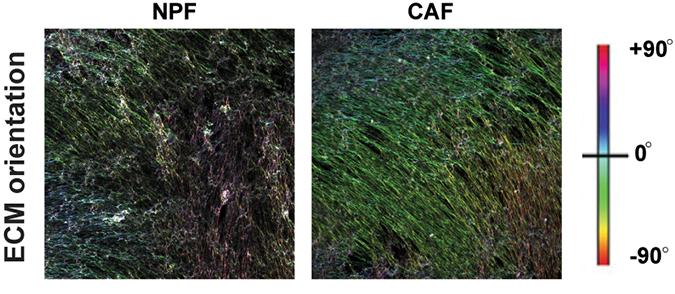 Cancer-associated fibroblasts can alter cellular organization in prostate cancer (right) versus healthy tissue (left).Elizabeth V. Nguyen et al./MCP
Cancer-associated fibroblasts can alter cellular organization in prostate cancer (right) versus healthy tissue (left).Elizabeth V. Nguyen et al./MCP
A hormonal link between diet and obesity
Previous studies in mice have implicated the hormone adropin in metabolic homeostasis, but its role in the metabolism of humans is unknown. To gain a better understanding of adropin activity, Andrew Butler and colleagues at multiple institutions in the U.S. used the transcriptome atlas of a nonhuman primate to profile the expression of the adropin-encoding gene ENHO. A second nonhuman primate model of high-sugar diet indicated that low circulating adropin is predictive of increased weight gain and prediabetes. The authors suggest that the data, published in the Journal of Biological Chemistry, warrant the investigation of adropin administration to promote metabolic health.
Brain over binge: Unlocking metabolic obesity-related gender disparities
Diet-induced obesity is a global problem. Hypothalamic regulation, responsible for metabolic processes, and its relationship to obesity have become a promising avenue for research. The endocannabinoid system, found in the hypothalamus, also plays a role in metabolic functions, working like locks and keys, where cannabinoid receptors are the locks and the endocannabinoid proteins behave as keys, creating a positive feedback loop regulating energy homeostasis.
In a paper in the Journal of Lipid Research, Cristina Miralpeix and a team of researchers in Spain write that they have found a link between specific endocannabinoids and diet-induced obesity in mice while noting disparities between genders. Groups of male and female mice were put on a high-fat diet and observed over time. Endocannabinoid levels of all the mice increased four to six times as compared with initial levels after short-term changes in diet. The increase of transient endocannabinoids affected metabolic functions such as body heat and weight gain. Furthermore, female mice became obese later than males, and their hypothalamic endocannabinoids showed higher levels, suggesting not only differences between sexes but also an inverse relationship between weight and endocannabinoid levels. The results also showed a striking rise in endocannabinoids after a short-term high-fat diet in all the mice that leveled off when a high-fat diet was maintained long-term.
This study could add insight into the relationship between hypothalamic regulation and obesity, and the results support claims of differences in male and female metabolic tendencies.
How disease and diet influence transcription
The Mediator complex is a multiprotein assembly and transcriptional coactivator that connects transcription factors to RNA polymerase II. Past research suggested that the Mediator complex can come in two sizes, but it has not been clear what determines the form of the assembly. Dou Yeon Youn and researchers from Albert Einstein College of Medicine and Rutgers Robert Wood Johnson Medical School found that the large complex exists in the livers of fasted mice and that, after feeding, nutrient activation of the mTORC1 pathway converts the complex into a smaller form. They also found that insulin-resistant mice in the fasted state exhibited increased expression of the small Mediator complex. Taken together, these findings, published in the Journal of Biological Chemistry, demonstrate the dynamic regulation of the Mediator complex under physiological and pathophysiological conditions.
Cholesterol regulation and Alzheimer’s disease
Alzheimer’s disease, affecting more than 40 million people worldwide, destroys mental cognition as well as memory. Recent insight into maintenance of cholesterol homeostasis appears to offer answers to many of the questions posed by Alzheimer’s. About 30% of a person’s cholesterol is stored in the brain; when properly regulated, it plays an important role in neural activity through synaptogenesis and maintenance of plasticity and brain function. Not surprisingly, disruption of cholesterol homeostasis has been linked to neurodegenerative diseases such as Alzheimer’s.
Cinzia Marchi at the University of Parma and colleagues in Italy investigated the link between dysfunctional chemical cues from the cerebrospinal fluid to neural synapsing and Alzheimer’s disease. Their research was published in the Journal of Lipid Research. The team explored how the ability of cholesterol to be released from cells, known as cholesterol efflux, in cerebral spinal fluid was reduced in Alzheimer’s disease. Cholesterol efflux levels of Alzheimer’s patients were compared with healthy controls and people with dementia not caused by Alzheimer’s disease. The team studied the ABCA1 and ABCG1 genes, which are responsible for cholesterol traffic from the cerebrospinal fluid and known to be risk factors for Alzheimer’s when mutated.
To detect functioning of these genes, the researchers studied three biomarkers. In patients with Alzheimer’s, significantly less of each biomarker was produced during efflux. However, these markers were not compromised in subjects with dementia unrelated to Alzheimer’s. Furthermore, when apolipoprotein E, a known risk factor of Alzheimer’s, and apolipoprotein A-1 levels were studied, the former was lower in non-Alzheimer’s dementia, while the latter was comparable for all groups.
This investigation shows that the capacity to promote cell cholesterol efflux by ABCA1 and ABCG1 genes from cerebrospinal fluid is impaired in Alzheimer’s. These findings could lead to development of new lipoprotein-based pharmaceuticals for treatment of the disease.
— Tori Zirul
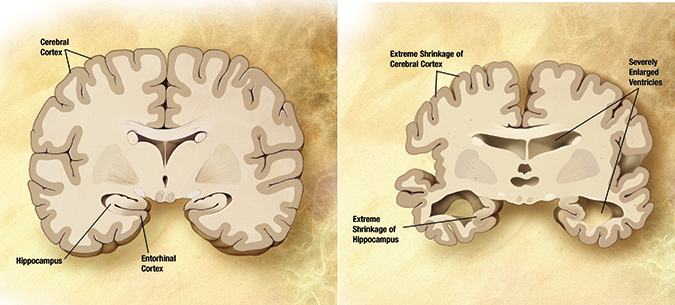 This illustration shows a cross section of a healthy brain, at left, and a brain affected by Alzheimer’s disease, at right.Alzheimer’s Disease Education and Referral Center.
This illustration shows a cross section of a healthy brain, at left, and a brain affected by Alzheimer’s disease, at right.Alzheimer’s Disease Education and Referral Center.
Boosting essential and versatile coenzyme NAD+
Recent work has indicated that increased concentrations of the coenzyme nicotinamide adenine dinucleotide, or NAD+, may provide an assortment of health benefits, including protection from liver disease and neurodegeneration. As such, there has been a surge of interest in agents that can raise NAD+ concentrations. Several NAD+ precursors have been identified, but high doses are required to obtain health benefits in animal models.
Yue Yang and colleagues at Weill Cornell Medical College investigated the ability of dihydronicotinamide riboside, or NRH, a likely NAD+ precursor, to enhance NAD+ concentrations.
The researchers report in the Journal of Biological Chemistry that, compared with two well-studied NAD+ precursors, NRH was more effective at raising NAD+ concentration in mammalian cells and in mice.
A model for the proteomics of plant defense
Just as animal bodies repair themselves after injuries, so too do plants. Previous research has identified the protein systemin as a key driver for plant defense and wound healing. Systemin exerts its effects by binding to receptors on plant cell membranes and signaling for initiation of a defensive response, but the specifics of this process are poorly understood. A new study by Fatima Haj Ahmad and colleagues at the University of Hohenheim, published in the journal Molecular & Cellular Proteomics, helps elucidate systemin’s downstream effects.
Ahmad and her team treated tomato cells with systemin or an inactive systemin mutant and compared how the prevalence of phosphate groups in various proteins changed with time. By tracking these changes, the researchers identified over 50 kinases (phosphate adders) and phosphatases (phosphate removers) specifically affected by systemin signaling. This enabled them to create the most advanced model to date for the downstream effects of systemin and the defensive response in plants. This research provides a baseline for further elucidation of the systemin pathway and could provide crucial insights for botanists and farmers to help ensure plant health and defenses against damage and disease.
Enjoy reading ASBMB Today?
Become a member to receive the print edition monthly and the digital edition weekly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Cows offer clues to treat human infertility
Decoding the bovine reproductive cycle may help increase the success of human IVF treatments.
Immune cells can adapt to invading pathogens
A team of bioengineers studies how T cells decide whether to fight now or prepare for the next battle.
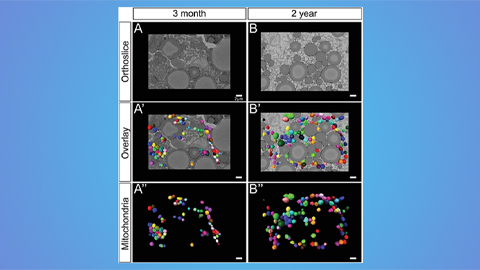
Hinton lab maps structure of mitochondria at different life stages
An international team determines the differences in the 3D morphology of mitochondria and cristae, their inner membrane folds, in brown adipose tissue.

National Academies propose initiative to sequence all RNA molecules
Unlocking the epitranscriptome could transform health, medicine, agriculture, energy and national security.
From the journals: JLR
What can you do with artificial lipoproteins? A new key to angiogenesis. Flavonoids counteract oxidative stress. Read about recent papers on these topics.
Iron could be key to treating a global parasitic disease
A study has found that leishmaniasis causes body-wide changes in iron balance, leading to red blood cell damage.



