JBC: A phospholipid pathway from plants to parasites
Findings by researchers at Washington University in St. Louis may aid in the development of therapies to treat parasitic infections, including malaria, and may help plant scientists one day produce hardier crops. The research team’s work was published in the Journal of Biological Chemistry.
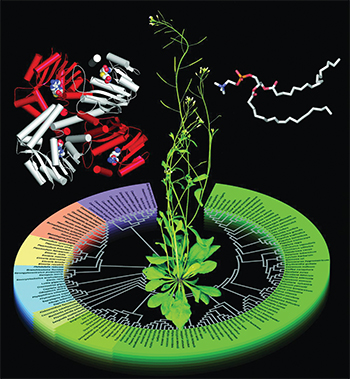 A study explains how structures of Arabidopsis phosphoethanolamine methyltransferase, or PMT, (left) are evolutionarily related to PMT sequences from different organisms. Phosphatidylcholine (right) is PMT's product. Courtesy of Soon Goo Lee and Joseph JezCholine is an essential nutrient that we get from certain foods, including eggs, meat, leafy greens and nuts. The human body converts choline into phosphocholine, or pCho, which it in turn converts into (among other essential building blocks) phosphatidylcholine, or PtdCho, a component of cell membranes. Plants can’t acquire the nutrient from the environment and so must synthesize pCho from scratch. The biochemical pathway plants use to synthesize pCho also is found in nematodes and the malaria parasite Plasmodium.
A study explains how structures of Arabidopsis phosphoethanolamine methyltransferase, or PMT, (left) are evolutionarily related to PMT sequences from different organisms. Phosphatidylcholine (right) is PMT's product. Courtesy of Soon Goo Lee and Joseph JezCholine is an essential nutrient that we get from certain foods, including eggs, meat, leafy greens and nuts. The human body converts choline into phosphocholine, or pCho, which it in turn converts into (among other essential building blocks) phosphatidylcholine, or PtdCho, a component of cell membranes. Plants can’t acquire the nutrient from the environment and so must synthesize pCho from scratch. The biochemical pathway plants use to synthesize pCho also is found in nematodes and the malaria parasite Plasmodium.
In plants, the enzymatic reaction that produces pCho is essential for normal function and for responding to stresses. Plant pCho is converted into PtdCho, which builds membranes that can adjust their rigidity in response to temperature changes. pCho also gets converted into molecules that help plants survive high salt. The enzymes that produce plant pCho are called phosphoethanolamine methyltransferases, or PMTs.
Soon Goo Lee, a postdoctoral research fellow at Washington University in the lab of Joseph Jez (a JBC associate editor), has been fascinated by PMTs in both plants and parasites for many years.
“Understanding the PMT enzyme is key to engineer plants with improved stress tolerance and enhanced nutrients,” Lee said.
Since the PMT-catalyzed pathway is found in parasites but not humans, Lee and Jez’s team is looking for inhibitors of this enzyme to treat diseases caused by these parasites.
The new study explains that PMTs of the model plant Arabidopsis thaliana share core features of PMTs from parasites, with almost identical structure at the active site. But the plant PMTs are roughly twice as large as the parasite ones, with large sections that can rearrange themselves to carry out multiple chemical reactions.
The three PMT types in the plant — which were thought to carry out the same function — actually appear to play different roles depending on where they are found in the plant. Plant growth experiments showed that one type of PMT was essential for root development and salt tolerance, whereas the other two had no effect on roots and instead seemed to be found primarily in leaves.
In the long run, this big-picture view of PMTs in different organisms offers routes to engineer enzymes with different functions. “I love these kinds of stories,” Lee said, “where I can look from the atomic (structure) to the physiological level to explain why these enzymes have different forms and how they work.”
Enjoy reading ASBMB Today?
Become a member to receive the print edition monthly and the digital edition weekly.
Learn moreGet the latest from ASBMB Today
Enter your email address, and we’ll send you a weekly email with recent articles, interviews and more.
Latest in Science
Science highlights or most popular articles
Iron could be key to treating a global parasitic disease
A study has found that leishmaniasis causes body-wide changes in iron balance, leading to red blood cell damage.

Environmental DNA is everywhere
The ability to extract trace bits of DNA from soil, water, and even air is revolutionizing science. Are there pitfalls?
Early COVID-19 research is riddled with poor methods and low-quality results
The pandemic worsened, but didn’t create, this problem for science.

From the journals: MCP
Three views of mass spec: analyzing secreted protein spectra, imaging mass spectrometry for clinical use and spectral libraries for MS data analysis. Read about these recent papers.

Understanding the fat science
Researchers at UCLA investigate lipid remodeling in the liver for energy generation.

No oxygen? No problem
By studying how electric fish survive in hypoxic streams for months at time, researchers may find new ways to target tumors.

.jpg?lang=en-US&width=300&height=300&ext=.jpg)